XML Backbone
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
What Is XML Backbone?
The XML Backbone is the core structural component of an electronic Common Technical Document (eCTD) submission. It acts as the electronic table of contents that organizes, identifies, and links every document within the eCTD dossier.
Definition
XML (Extensible Markup Language) is a standardized markup language used to encode data in a structured and machine-readable format. In eCTD, the XML backbone provides metadata and structural information that allows regulatory authorities to interpret the content and hierarchy of the submission.
Purpose of XML Backbone
The XML backbone does not contain the scientific data itself. Instead, it defines how the documents are arranged within Modules 1 to 5 and specifies their relationships. It enables regulatory review systems to navigate through large volumes of documents efficiently.
Each document submitted in PDF format is referenced within the XML file, which includes details such as document title, file path, version, sequence number, and lifecycle operation.
Key Functions
The XML backbone performs the following critical functions:
It defines the folder structure according to the harmonized specifications developed by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use.
It assigns lifecycle operators such as new, replace, append, or delete to manage updates.
It organizes documents under the correct CTD section and subsection.
It supports hyperlinking and cross-referencing within the dossier.
It ensures compatibility with regulatory authority validation tools.
Lifecycle Management in XML
One of the most important roles of the XML backbone is lifecycle management. Each time a regulatory submission is updated, a new sequence number is generated. The XML file identifies whether documents are newly added or replacing previous versions. This allows authorities such as the U.S. Food and Drug Administration or the European Medicines Agency to track the complete submission history without resubmission of the entire dossier.
Technical Structure
An XML backbone includes:
Sequence number identifying each submission step.
Leaf elements representing individual documents.
Attributes defining document type and lifecycle operator.
Links to corresponding PDF files within the eCTD folder structure.
It is validated using regional technical validation criteria before submission to ensure compliance.
Importance in Regulatory Review
Without the XML backbone, the eCTD submission cannot function properly. It ensures structured navigation, accurate document placement, effective lifecycle tracking, and standardized electronic communication between pharmaceutical companies and regulatory authorities.
Conclusion
The XML backbone is the structural framework of eCTD. It transforms a collection of PDF documents into a structured, navigable, and lifecycle-managed electronic regulatory submission that meets global harmonized standards.
make tables wherever possible
make tables wherever possible
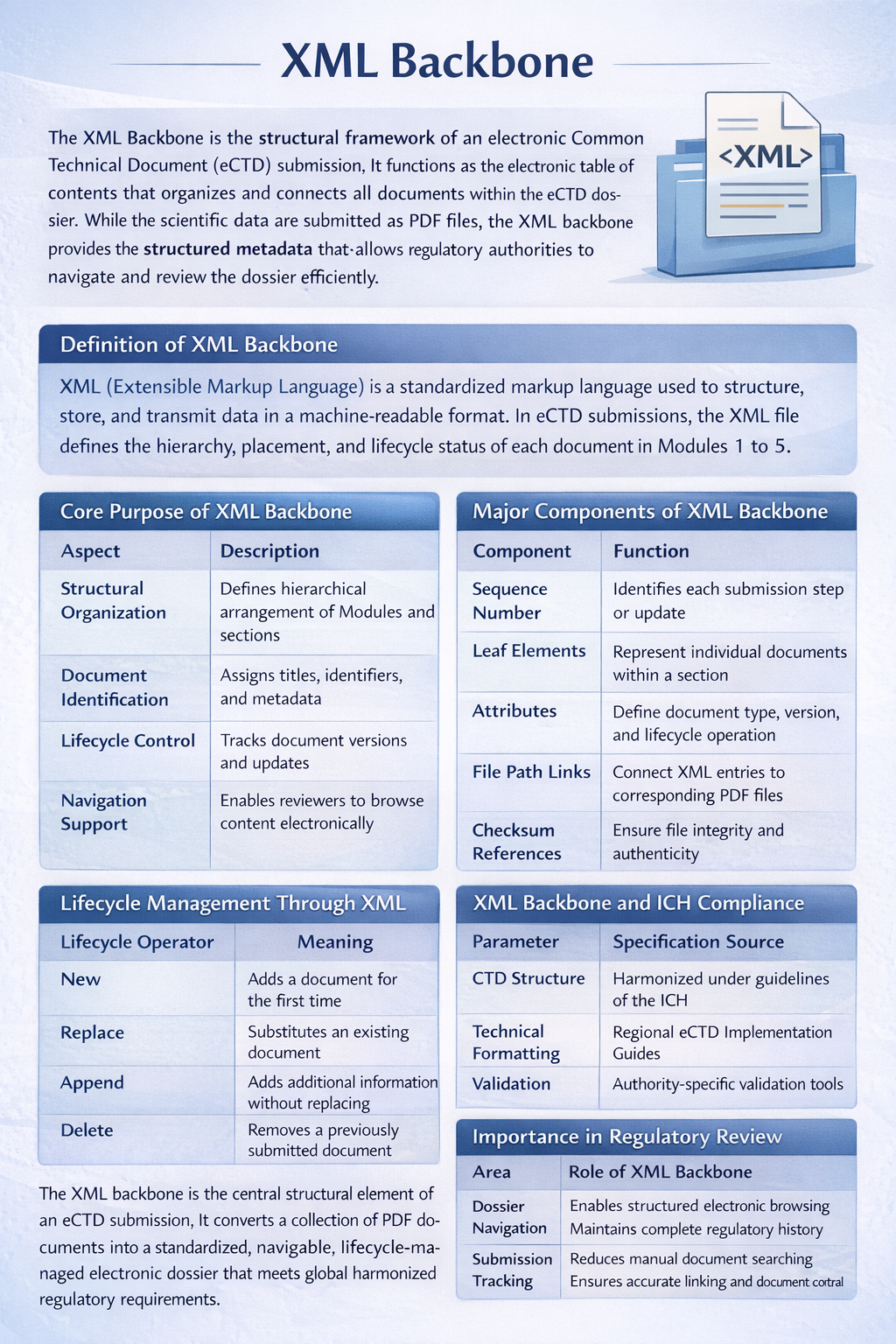
XML Backbone
The XML Backbone is the structural framework of an electronic Common Technical Document (eCTD) submission. It functions as the electronic table of contents that organizes and connects all documents within the eCTD dossier. While the scientific data are submitted as PDF files, the XML backbone provides the structured metadata that allows regulatory authorities to navigate and review the dossier efficiently.
Definition of XML Backbone
XML (Extensible Markup Language) is a standardized markup language used to structure, store, and transmit data in a machine-readable format. In eCTD submissions, the XML file defines the hierarchy, placement, and lifecycle status of each document in Modules 1 to 5.
Core Purpose of XML Backbone
| Aspect | Description |
|---|---|
| Structural Organization | Defines hierarchical arrangement of Modules and sections |
| Document Identification | Assigns titles, identifiers, and metadata |
| Lifecycle Control | Tracks document versions and updates |
| Navigation Support | Enables reviewers to browse content electronically |
| Regulatory Validation | Ensures compliance with regional technical criteria |
Major Components of XML Backbone
| Component | Function |
|---|---|
| Sequence Number | Identifies each submission step or update |
| Leaf Elements | Represent individual documents within a section |
| Attributes | Define document type, version, and lifecycle operation |
| File Path Links | Connect XML entries to corresponding PDF files |
| Checksum References | Ensure file integrity and authenticity |
Lifecycle Management Through XML
One of the most significant features of the XML backbone is lifecycle management. Every new submission or update is assigned a unique sequence number. Instead of resubmitting the entire dossier, only modified documents are submitted.
| Lifecycle Operator | Meaning |
|---|---|
| New | Adds a document for the first time |
| Replace | Substitutes an existing document |
| Append | Adds additional information without replacing |
| Delete | Removes a previously submitted document |
This system enables regulatory authorities such as the U.S. Food and Drug Administration and the European Medicines Agency to track submission history efficiently.
XML Backbone and ICH Compliance
| Parameter | Specification Source |
|---|---|
| CTD Structure | Harmonized under guidelines of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use |
| Technical Formatting | Regional eCTD Implementation Guides |
| Validation Criteria | Authority-specific validation tools |
Importance in Regulatory Review
| Area | Role of XML Backbone |
|---|---|
| Dossier Navigation | Enables structured electronic browsing |
| Submission Tracking | Maintains complete regulatory history |
| Review Efficiency | Reduces manual document searching |
| Data Integrity | Ensures accurate linking and document control |
Conclusion
The XML backbone is the central structural element of an eCTD submission. It converts a collection of PDF documents into a standardized, navigable, lifecycle-managed electronic dossier that meets global harmonized regulatory requirements.
