Types of IND
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
In the United States regulatory framework governed by the US Food and Drug Administration under 21 CFR Part 312, Investigational New Drug Applications (INDs) are categorized based on the sponsor’s intent and the regulatory pathway being pursued. Understanding the different types of INDs is essential for any Drug Regulatory Affairs professional, as each type carries distinct compliance responsibilities, documentation requirements, and strategic implications.
The primary classification of INDs includes Commercial IND, Investigator IND, and Emergency Use IND. These categories are defined by sponsorship structure and development objective, and they determine how regulatory oversight is applied.
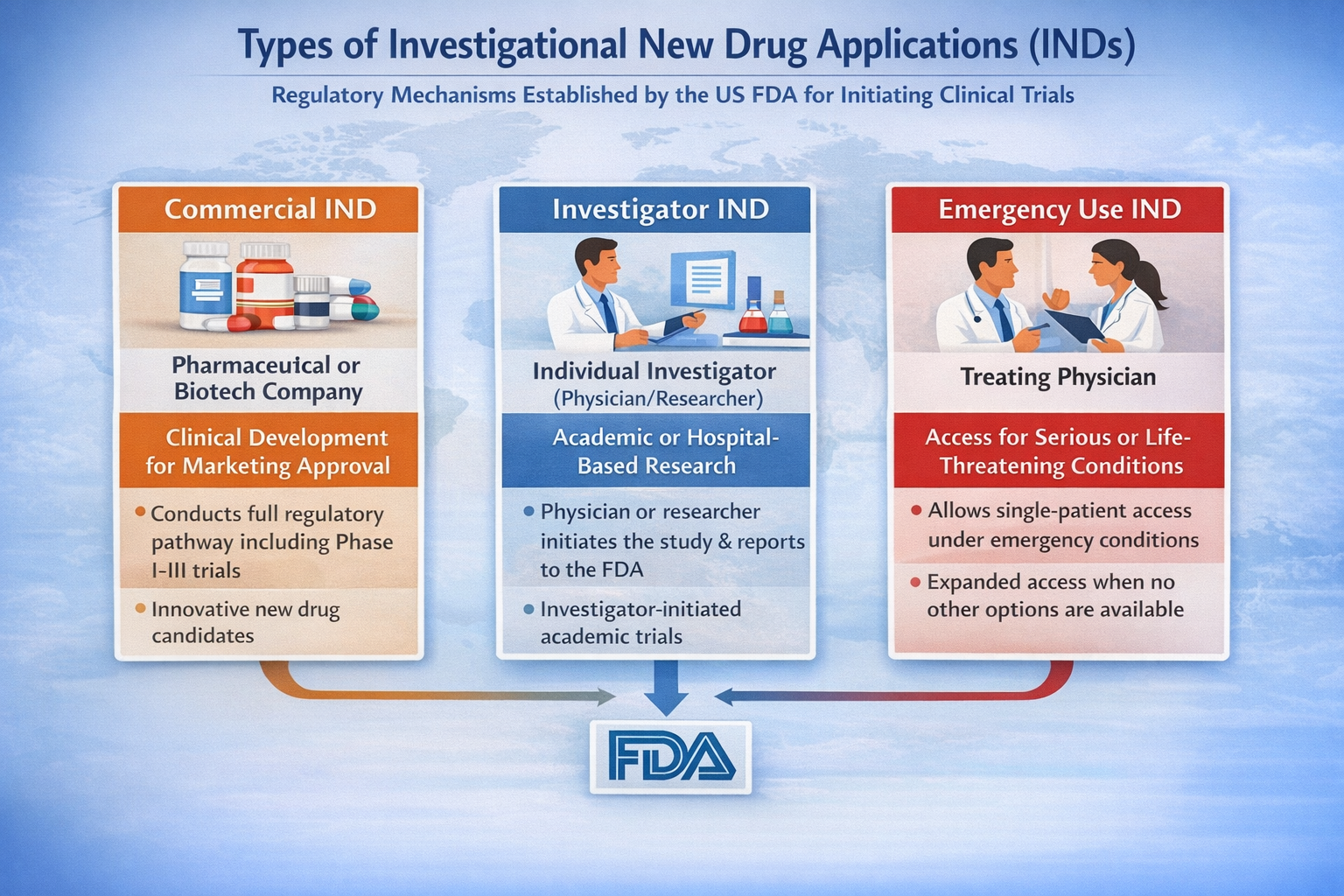
| IND Type | Sponsor | Primary Objective | Regulatory Context | Typical Industry Scenario |
|---|---|---|---|---|
| Commercial IND | Pharmaceutical or Biotechnology Company | Support clinical development leading to marketing approval | Full regulatory development pathway | Phase I–III trials for innovative drugs |
| Investigator IND | Individual Investigator (Physician/Researcher) | Conduct investigator-initiated research | Academic or institutional oversight | Hospital-based or academic clinical research |
| Emergency Use IND | Treating Physician | Provide investigational drug in urgent, life-threatening situations | Single-patient access under emergency conditions | Compassionate use where no alternatives exist |
A Commercial IND is the most common and strategically significant type in the pharmaceutical industry. It is typically filed by a sponsor company intending to develop a new drug for eventual marketing approval through a New Drug Application (NDA) or Biologics License Application (BLA). This IND type requires comprehensive documentation covering nonclinical studies, Chemistry, Manufacturing and Controls (CMC), and detailed clinical protocols. Regulatory professionals working in multinational companies such as Pfizer, Novartis, Roche, or IQVIA are primarily involved in Commercial IND submissions. These INDs follow a structured development plan that includes multiple protocol amendments, safety reports, and annual reports throughout the lifecycle of clinical development.
An Investigator IND, on the other hand, is submitted by an individual physician who both initiates and conducts the clinical investigation. In this case, the investigator assumes the dual role of sponsor and clinical investigator, making them responsible for regulatory compliance, safety reporting, and record maintenance. Investigator INDs are common in academic research settings, where physicians may study new indications, dosing regimens, or combinations of existing drugs. Although less commercially driven, the regulatory standards for safety and documentation remain stringent. For regulatory professionals supporting academic institutions, advisory roles often involve ensuring adherence to IND maintenance requirements and ethical oversight.
The Emergency Use IND is a special regulatory mechanism designed for urgent situations involving serious or life-threatening conditions where no satisfactory therapeutic alternatives exist. This pathway allows a physician to obtain authorization from the FDA to use an investigational drug for a single patient outside of a clinical trial. In emergency circumstances, authorization may initially be granted verbally by the FDA, followed by formal written documentation. This type of IND reflects the balance between regulatory control and patient access to potentially life-saving therapies. It is often associated with expanded access or compassionate use programs.
In addition to the three primary types, regulatory professionals must also understand sub-classifications that influence compliance strategy. Treatment INDs and Expanded Access INDs are variations that permit broader patient access to investigational drugs outside traditional clinical trials. These are typically used during late-stage clinical development when substantial evidence of safety and efficacy is available but marketing approval has not yet been granted.
| Special Category | Purpose | Development Stage | Regulatory Consideration |
|---|---|---|---|
| Treatment IND | Provide access to promising drugs | Late Phase II or Phase III | Requires sufficient safety and efficacy data |
| Expanded Access IND | Compassionate use for groups of patients | During clinical development | Strict safety monitoring required |
From a job-readiness perspective, understanding the distinctions between these IND types is critical because documentation strategy, risk assessment, and regulatory interactions differ significantly. Commercial INDs require long-term lifecycle planning, while Investigator INDs demand strong compliance oversight from smaller research settings. Emergency and Expanded Access INDs require rapid regulatory communication and clear ethical justification.
Strategically, regulatory professionals must assess the development goal before determining the appropriate IND type. A company planning global commercialization will pursue a Commercial IND with a structured clinical development plan aligned with FDA expectations. An academic investigator exploring a novel therapeutic hypothesis may pursue an Investigator IND with limited scope. In urgent medical cases, physicians may rely on Emergency Use INDs to provide timely patient care.
In conclusion, the types of INDs reflect different regulatory intentions, sponsorship models, and patient access mechanisms within the United States drug development system. Mastery of these distinctions enables Regulatory Affairs professionals to select the appropriate pathway, ensure compliance, and align regulatory strategy with scientific and commercial objectives.
