GxP Overview
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
GxP Overview – Foundation of Pharmaceutical Quality and Regulatory Compliance
In the pharmaceutical and life sciences industry, GxP represents the collective framework of quality guidelines and regulations that ensure pharmaceutical products are consistently safe, effective, and manufactured to the highest standards. The “G” stands for “Good,” and the “x” represents different regulated practices such as Manufacturing, Clinical, Laboratory, and Distribution. Mastery of GxP principles is essential for professionals entering Regulatory Affairs, Quality Assurance, Clinical Research, Manufacturing, or Pharmacovigilance. Without a strong understanding of GxP, it is impossible to function effectively in a regulated pharmaceutical environment.
GxP regulations are enforced globally by authorities such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and India’s Central Drugs Standard Control Organization (CDSCO). Although regional requirements may vary, the core objective remains consistent: protecting patient safety and ensuring data integrity. GxP compliance is grounded in three fundamental pillars: product quality, patient safety, and data integrity. Every regulatory submission, inspection, audit, and approval decision ultimately depends on whether these pillars are maintained throughout the product lifecycle.
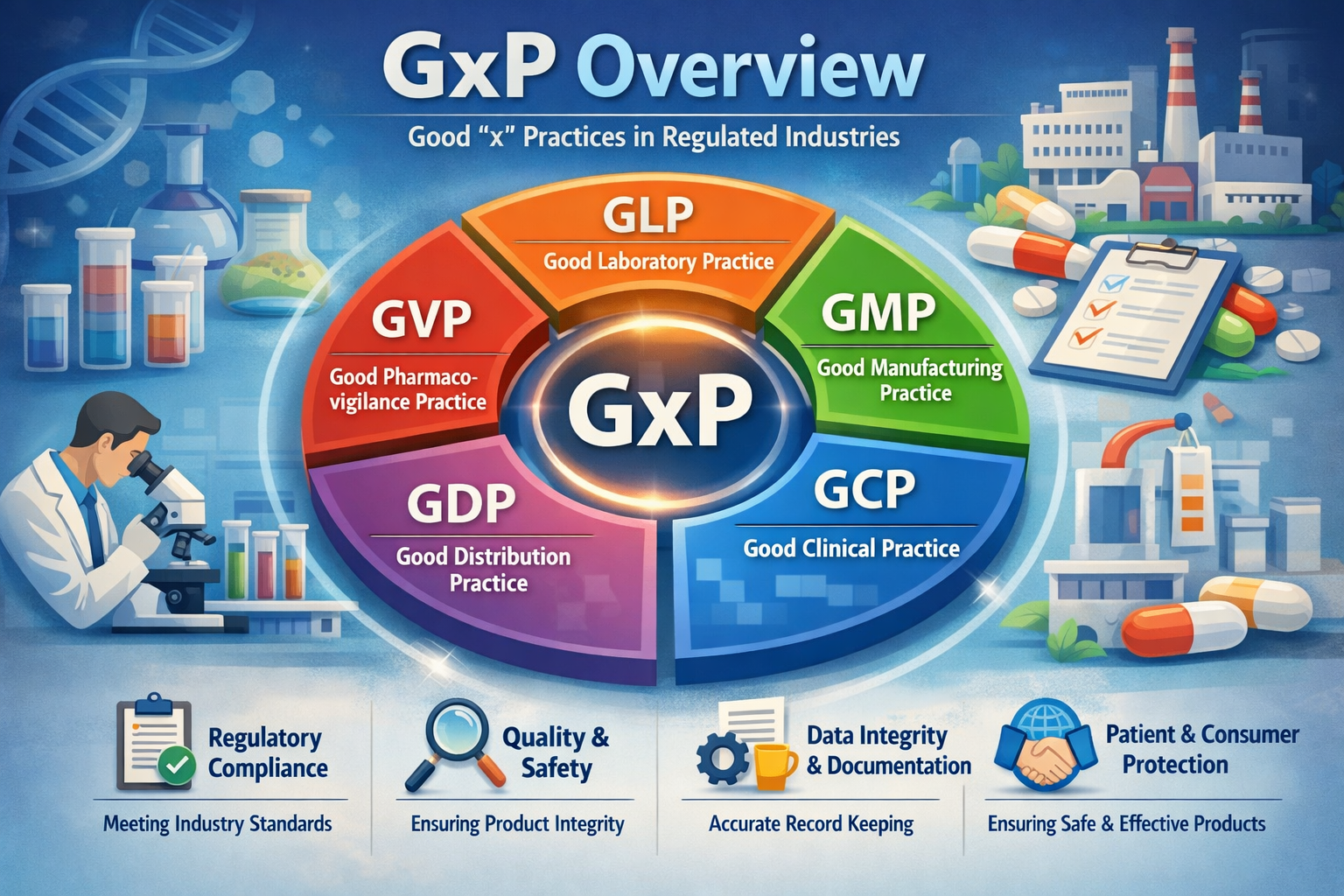
| GxP Domain | Scope | Key Applications | Career Relevance |
|---|---|---|---|
| Good Manufacturing Practice (GMP) | Governs manufacturing, processing, packaging, and storage of pharmaceutical products | Equipment and process validation, batch records, environmental monitoring, CAPA | Regulatory dossier preparation, CTD Module 3, inspection readiness |
| Good Clinical Practice (GCP) | Ensures clinical trial integrity and patient safety | Trial design, conduct, monitoring, reporting, investigator compliance | IND/NDA/MAA submissions, clinical data verification |
| Good Laboratory Practice (GLP) | Applies to non-clinical laboratory studies | Toxicology, pharmacology, method validation, documentation | Nonclinical data submission, preclinical study oversight |
| Good Distribution Practice (GDP) | Ensures quality and integrity during distribution | Cold-chain management, traceability, storage conditions | Supply chain compliance, inspection readiness |
| Good Pharmacovigilance Practice (GVP) | Drug safety monitoring post-marketing | Adverse event reporting, risk management plans | Post-marketing surveillance, safety submissions |
| Good Documentation Practice (GDP) | Accurate, legible, traceable record keeping | SOPs, batch records, validation reports, audit trails | Data integrity, regulatory inspection readiness |
Regulatory Affairs professionals do not simply compile documents; they certify compliance. Every submission implicitly declares that all studies, manufacturing processes, and quality systems comply with applicable GxP standards. During inspections, agencies review evidence of compliance. Deficiencies may result in warning letters, import alerts, or product recalls. A job-ready professional must be able to identify which GxP applies to a given activity, interpret inspection observations such as FDA Form 483 findings, review audit reports, evaluate validation protocols and summary reports, assess data integrity risks, and determine compliance risks prior to submission.
Data integrity is central to GxP compliance. Regulatory authorities require adherence to ALCOA principles: data must be Attributable, Legible, Contemporaneous, Original, and Accurate. Modern expectations extend to completeness, consistency, durability, and availability. For instance, in manufacturing, batch records must be completed in real time. Backdated entries or overwritten corrections constitute serious violations. In clinical trials, source documents must support all reported data, and in laboratory studies, raw data must be archived and retrievable. Understanding ALCOA ensures that regulatory submissions are accepted and that data withstand inspection scrutiny.
Documentation and audit readiness are key components of GxP compliance. All SOPs, training records, validation protocols, change controls, deviations, and CAPA reports must be properly maintained. Professionals must adopt an inspector’s mindset, asking whether processes are validated, evidence is documented, deviations are investigated, risk assessments are recorded, and personnel are appropriately trained and qualified. A lifecycle perspective is essential because GxP applies across all phases of drug development: GLP in discovery and preclinical research, GCP during clinical development, GMP during commercial manufacturing, GDP during distribution, and GVP in post-marketing safety monitoring. Regulatory strategy must integrate all these domains to ensure seamless product approval and lifecycle management.
| Product Lifecycle Stage | Applicable GxP | Key Compliance Activities |
|---|---|---|
| Discovery / Preclinical | GLP | Toxicology study documentation, method validation, study protocols |
| Clinical Development | GCP | Clinical trial design, patient safety monitoring, ethical approvals |
| Manufacturing / Production | GMP | Equipment validation, process control, batch record review, CAPA |
| Distribution / Supply Chain | GDP | Cold-chain management, traceability, storage compliance |
| Post-Marketing | GVP | Adverse event reporting, risk management plans, safety monitoring |
Practical career applications for beginners include reviewing GMP audit reports to understand common deficiencies, studying SOP structures and validation master plans, mapping GxP documentation into CTD modules, analyzing inspection findings for regulatory risk, and identifying compliance gaps through case studies. During interviews, scenario-based questions may include handling a deviation, responding to a warning letter, or ensuring data integrity during submission preparation. Strong conceptual understanding of GxP, combined with practical familiarity with audits, documentation, and lifecycle integration, enables confident and structured responses, preparing candidates to function effectively in regulated pharmaceutical environments.
Mastery of GxP principles is foundational for any career in pharmaceutical quality, regulatory affairs, clinical research, pharmacovigilance, or manufacturing. It ensures that professionals can navigate complex regulatory frameworks, maintain compliance throughout the product lifecycle, and contribute to patient safety and global healthcare standards.
