Regional Differences in Module 1
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
Introduction
Module 1 of the Common Technical Document (CTD) is unique in that it is not harmonized under ICH guidelines. While Modules 2 through 5 are globally standardized, Module 1 contains region-specific administrative and legal information required by regulatory authorities. The content and format of Module 1 vary depending on the jurisdiction, reflecting local regulations, submission procedures, and legal requirements. For Regulatory Affairs professionals, understanding these regional differences is critical for ensuring compliance, avoiding delays, and preparing submission-ready dossiers tailored to each market.
Purpose of Regional Differences
The main purpose of region-specific requirements in Module 1 is to ensure that regulatory authorities have all the administrative, legal, and procedural information necessary to process a submission. These requirements allow authorities to confirm the applicant’s identity, authority, and compliance with local laws, and to verify that the submitted product meets regional standards. For professionals, adapting Module 1 content to local requirements is essential for successful submissions, as errors or omissions in administrative documentation are a common reason for delays or rejection.
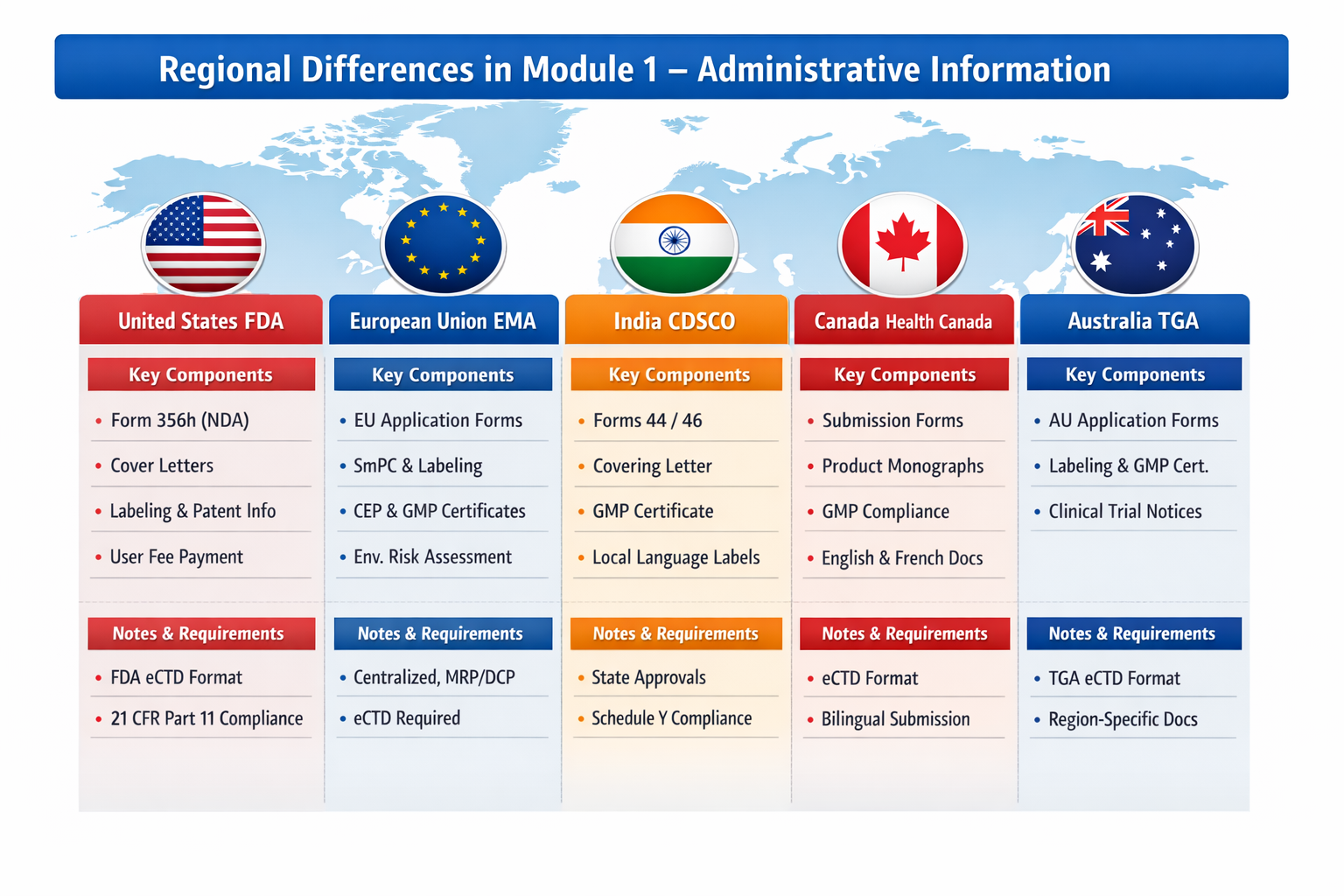
Regional Module 1 Requirements
| Region | Module 1 Key Components | Notes / Special Requirements |
|---|---|---|
| United States (FDA) | Form 356h (New Drug Application), cover letters, administrative checklists, labeling, patent certifications, user fee payment information, orphan drug status (if applicable) | Must comply with FDA eCTD specifications; submission fees must be included; electronic signatures are validated under 21 CFR Part 11. |
| European Union (EMA) | Regional application forms, cover letter, product information (SmPC, labeling), Certificates of Suitability (CEP) for drug substances, GMP certificates, environmental risk assessments | Submission format depends on centralized, decentralized, or mutual recognition procedures; specific electronic submission requirements for eCTD are strictly enforced. |
| India (CDSCO) | Application forms (Form 44, 46), covering letter, labeling, GMP certificate, test license, manufacturing license, product composition details | Local language labeling may be required; CDSCO requires approvals from State Licensing Authorities; alignment with Schedule Y regulations is essential. |
| Canada (Health Canada) | Submission forms, cover letters, labeling, product monographs, GMP compliance, Certificates of Analysis | Health Canada requires Common Technical Document Module 1 documents in both English and French; electronic submissions follow eCTD specifications. |
| Australia (TGA) | Application forms, cover letters, labeling, GMP certificate, clinical trial notifications (if applicable) | TGA requires region-specific forms even when dossier follows ICH CTD structure; eCTD mandatory for most submissions. |
Practical Application for Regulatory Affairs Professionals
Understanding regional differences in Module 1 is essential for multi-region submissions. Regulatory Affairs teams must ensure that the correct forms, certificates, and legal documents are included for each jurisdiction. This requires close coordination with cross-functional teams, such as legal, quality, and clinical operations, to verify signatures, payment of fees, GMP compliance, and labeling standards. Preparing a region-specific Module 1 checklist for each authority helps prevent omissions and ensures submission efficiency. Failure to comply with regional Module 1 requirements is a common cause of administrative rejection or major regulatory queries.
Challenges and Best Practices
The primary challenge is managing the variability of regional requirements while maintaining consistency with Modules 2 through 5. Best practices include maintaining updated regulatory intelligence, using region-specific templates, validating documents before submission, and keeping detailed records of correspondence and approvals. It is also important to understand electronic submission requirements, as authorities such as the FDA, EMA, and TGA enforce strict eCTD standards. Cross-referencing Module 1 content with supporting data in other modules ensures completeness and avoids regulatory discrepancies.
Conclusion
Module 1 is the most region-specific part of the CTD, and its content must be tailored to meet the administrative and legal requirements of each regulatory authority. Regulatory Affairs professionals must master these regional differences to prepare compliant, complete, and submission-ready dossiers. Understanding local forms, certifications, labeling, and procedural nuances reduces delays, minimizes queries, and ensures smooth regulatory review. Mastery of Module 1 regional requirements is therefore essential for job-ready professionals managing global pharmaceutical submissions.
