eCTD Overview
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
What Is eCTD?
The electronic Common Technical Document (eCTD) is the standardized electronic format used for submission of regulatory dossiers to health authorities for marketing authorization of pharmaceutical products. It is based on the Common Technical Document framework developed by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use under the ICH M4 guideline.
eCTD does not change the scientific content of the dossier; instead, it defines the electronic structure, format, and technical standards for submission. It enables pharmaceutical companies to submit applications for new drug approvals, generic drug approvals, variations, renewals, and lifecycle updates in a harmonized electronic format.
Purpose of eCTD
The primary purpose of eCTD is to harmonize regulatory submissions across different regions while improving efficiency in review and lifecycle management. It replaces traditional paper submissions and allows regulatory authorities to navigate, review, and track changes more effectively using specialized electronic systems.
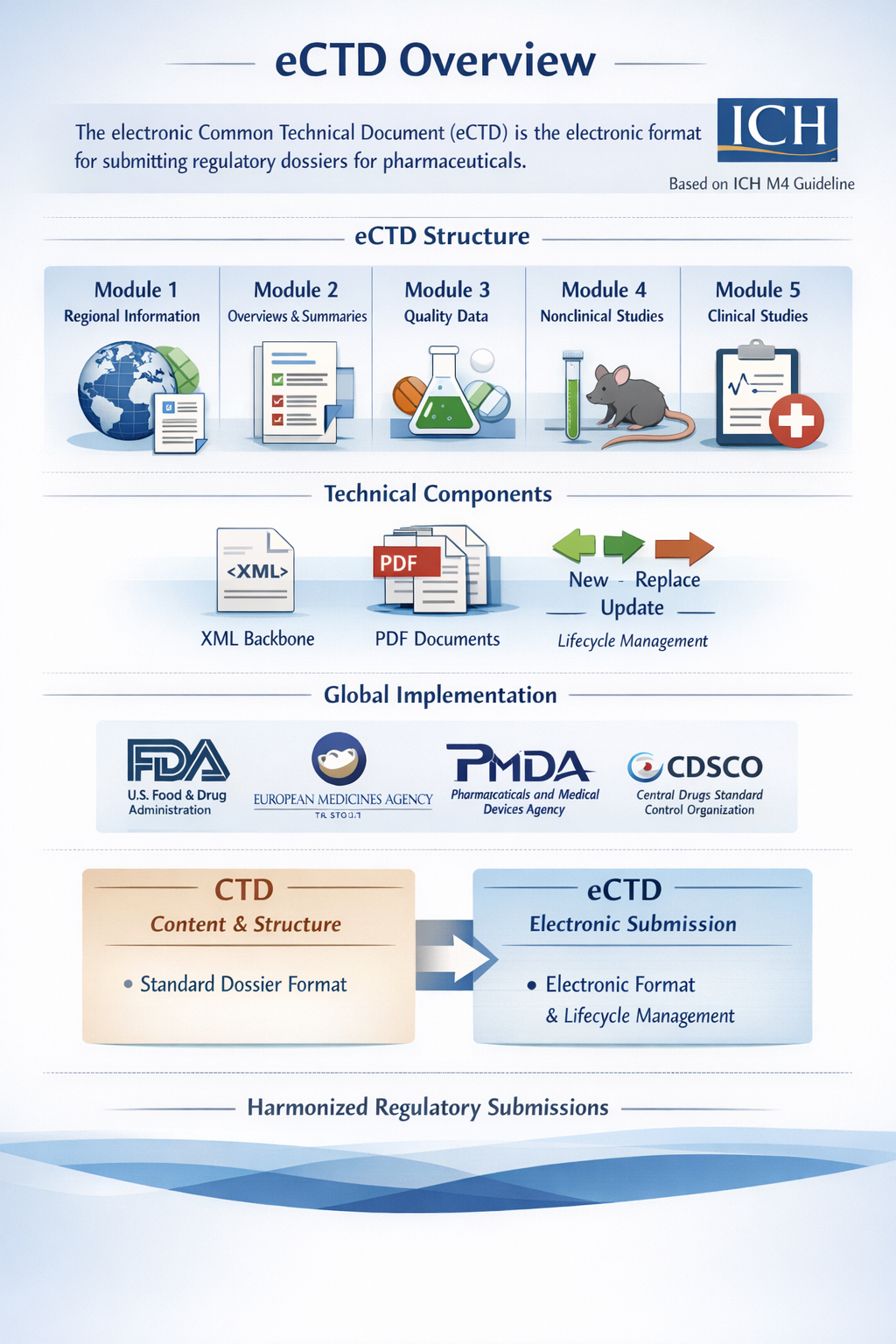
eCTD Structure
The eCTD follows the same five-module structure as the CTD:
Module 1 contains region-specific administrative information such as application forms, prescribing information, labeling, and country-specific regulatory requirements. The content of Module 1 varies according to the regulatory authority.
Module 2 contains high-level summaries of the dossier including the Quality Overall Summary, Non-clinical Overview and Summary, and Clinical Overview and Summary. This module provides a critical evaluation of the detailed data presented in later modules.
Module 3 includes quality documentation related to the drug substance and drug product. It provides detailed information about manufacturing processes, control of materials, specifications, validation, and stability studies.
Module 4 contains non-clinical study reports such as pharmacology, pharmacokinetics, and toxicology studies conducted in animals.
Module 5 includes clinical study reports such as Phase I, Phase II, and Phase III clinical trials, bioequivalence studies, and other clinical data supporting safety and efficacy.
Technical Components of eCTD
An eCTD submission consists of an XML backbone file and organized PDF documents structured in a predefined folder hierarchy. The XML backbone acts as an electronic table of contents that links and organizes all documents within the dossier. Each document is submitted in accordance with regional technical specifications.
A key feature of eCTD is lifecycle management. Instead of resubmitting the entire dossier for minor changes, applicants submit only updated documents using lifecycle operators such as new, replace, append, or delete. This allows regulatory authorities to track the history of changes systematically.
Global Implementation
eCTD is mandatory in many major regulatory regions including the U.S. Food and Drug Administration, the European Medicines Agency, and Japan’s Pharmaceuticals and Medical Devices Agency. In India, the Central Drugs Standard Control Organization has also adopted electronic submission systems aligned with global standards.
Difference Between CTD and eCTD
CTD refers to the standardized content and format of the technical documentation.
eCTD refers to the electronic format, technical structure, and submission mechanism used to present the CTD content to regulatory authorities.
Conclusion
eCTD is the globally accepted electronic standard for pharmaceutical regulatory submissions. It enhances harmonization, improves review efficiency, supports structured lifecycle management, and ensures consistent communication between applicants and regulatory agencies.
