CDSCO Structure and Indian Approval Process
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
Overview
The Central Drugs Standard Control Organization (CDSCO) is the national regulatory authority of India responsible for regulating pharmaceuticals, medical devices, and clinical trials. It operates under the Ministry of Health and Family Welfare, Government of India. CDSCO ensures the safety, efficacy, and quality of drugs marketed in India in accordance with the Drugs and Cosmetics Act, 1940, and the associated regulatory rules.
The organization functions through a centralized regulatory framework led by the Drugs Controller General of India, with support from zonal offices and state drug control authorities. This structure enables coordinated oversight of drug approvals, manufacturing, imports, and post-marketing safety.
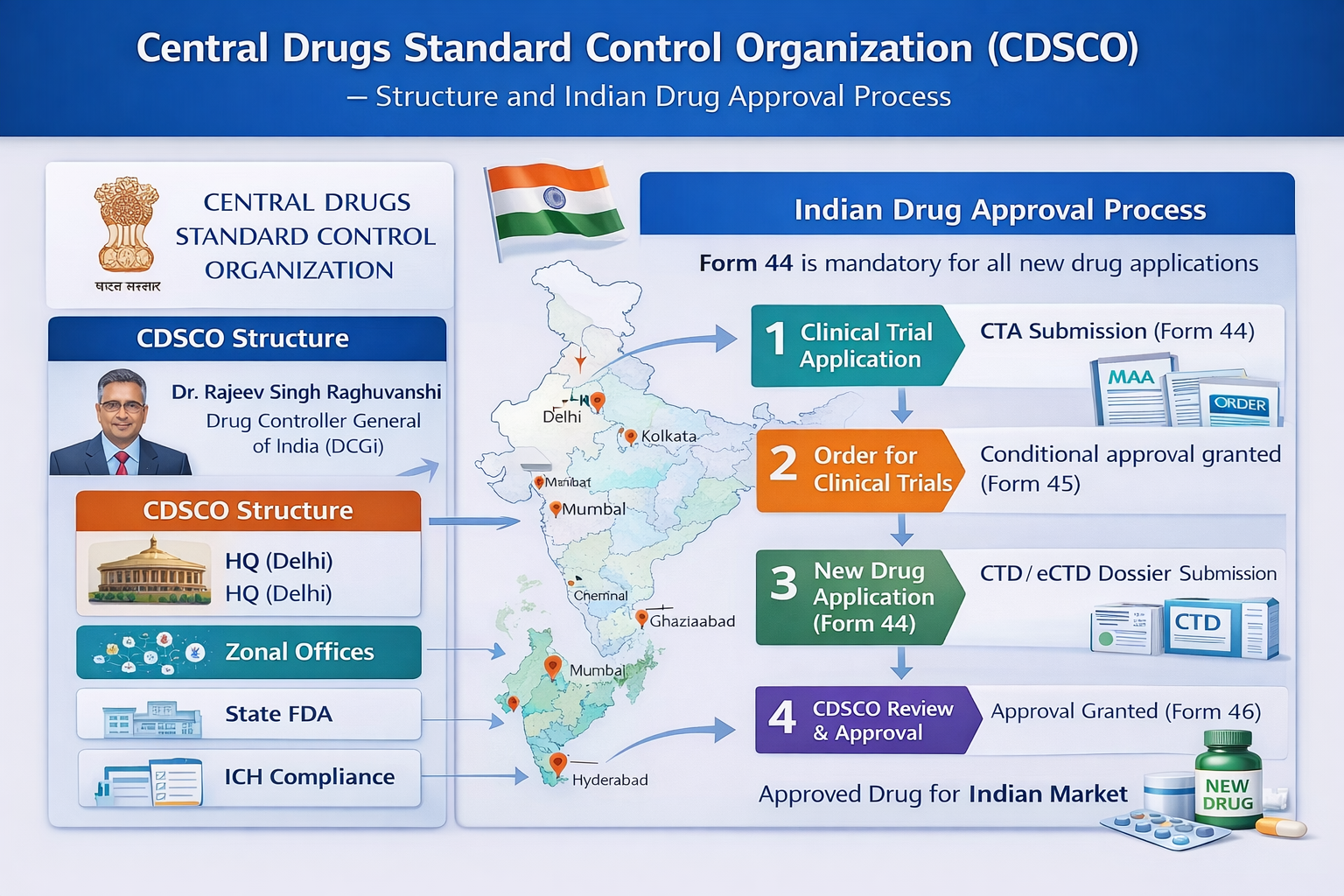
CDSCO Organizational Structure
CDSCO is headed by the Drugs Controller General of India (DCGI), who serves as the chief regulatory authority. The DCGI is responsible for approving new drugs and clinical trials, granting marketing authorizations, and regulating the import of pharmaceutical products into India. Supporting the DCGI are Joint Drugs Controllers, who oversee specialized regulatory divisions and assist in policy implementation.
Deputy and Assistant Drugs Controllers work under the senior leadership and handle technical evaluations of applications, review scientific data, and support regulatory decision-making. The CDSCO headquarters is located in New Delhi, where major activities such as policy formulation, evaluation of new drug applications, and clinical trial approvals are conducted.
To strengthen regulatory enforcement across the country, CDSCO operates through zonal and sub-zonal offices. These offices conduct inspections, monitor manufacturing and import units, and implement regulatory actions when required. Major zonal offices are located in cities such as Mumbai, Chennai, Kolkata, and Ghaziabad.
In addition to central operations, State Drug Control Authorities play a crucial role in the regulatory system. These state-level authorities grant manufacturing and sales licenses, conduct routine inspections of facilities, and oversee drug testing laboratories. They work in coordination with CDSCO to ensure consistent regulatory enforcement across India.
Regulatory decision-making is supported by expert advisory bodies. Subject Expert Committees review clinical trial and new drug applications and provide scientific recommendations. These recommendations are evaluated by the Technical Committee, which performs a higher-level review. The Apex Committee serves as the final decision-making authority for major regulatory approvals.
Indian Drug Approval Process
The drug approval pathway in India depends on the type of product, such as new drugs, generics, imported products, or biologics. However, the process for a new drug generally follows a structured sequence of regulatory steps.
The process begins with preclinical studies. These include laboratory and animal studies that evaluate the safety, toxicity, and pharmacological profile of the drug. The results of these studies form the basis for applying to conduct human clinical trials.
After successful preclinical testing, the sponsor submits a clinical trial application through the SUGAM online portal. This submission includes preclinical data, the clinical trial protocol, the investigator’s brochure, and ethics committee approval. The application is reviewed by the Subject Expert Committee to determine whether the trial can proceed.
Once approval is granted, clinical trials are conducted in three phases under the approved protocol. These trials must follow Good Clinical Practice guidelines and include mandatory safety monitoring and reporting of adverse events.
After successful completion of clinical trials, the sponsor submits a New Drug Application. This dossier includes complete quality data related to chemistry, manufacturing, and controls, as well as nonclinical and clinical study results, stability data, and proposed labeling. The application is reviewed sequentially by the Subject Expert Committee, the Technical Committee, and the Apex Committee.
If the application is approved, the DCGI grants marketing authorization for the drug. Following this, the State Drug Control Authority issues the manufacturing license, allowing the product to be produced and marketed in India.
Generic Drug Approval Process
For generic drugs, the approval process is typically shorter because the safety and efficacy of the reference product are already established. The main requirement is to demonstrate bioequivalence with the reference product. The applicant must submit a regulatory dossier that includes quality documentation and stability data to confirm shelf life and storage conditions. After regulatory review, CDSCO grants approval for the generic product.
Timelines
Clinical trial approvals generally take around ninety days if the documentation is complete and no major issues are identified. Marketing authorization timelines can vary depending on the complexity of the application, the number of review queries, and the completeness of the submitted data.
Post-Marketing Requirements
After a drug is approved and marketed, regulatory responsibilities continue. Companies must submit Periodic Safety Update Reports to monitor the ongoing safety of the product. Pharmacovigilance systems must be in place to report adverse events. In addition, manufacturing facilities are subject to regular compliance inspections to ensure adherence to Good Manufacturing Practices and quality standards.
Process Flow Summary
The overall drug approval process in India follows a structured sequence. It begins with preclinical studies, followed by submission of a clinical trial application. After approval, clinical trials are conducted. A New Drug Application is then submitted and reviewed by expert committees. Once the DCGI grants approval, the manufacturing license is issued by the state authority, allowing the product to be marketed.
CDSCO operates through a centralized leadership system supported by zonal and state authorities. Its structured approval pathway ensures scientific evaluation, regulatory oversight, and continuous post-marketing surveillance, helping safeguard public health across India.