TGA Australia Overview
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
Therapeutic Goods Administration (TGA) – Australia
The Therapeutic Goods Administration (TGA) is Australia’s national regulatory authority responsible for regulating medicines, biologics, and medical devices. It operates under the Department of Health and Aged Care and administers the Therapeutic Goods Act 1989. Its primary objective is to ensure that therapeutic goods supplied in Australia are safe, effective, and of high quality.
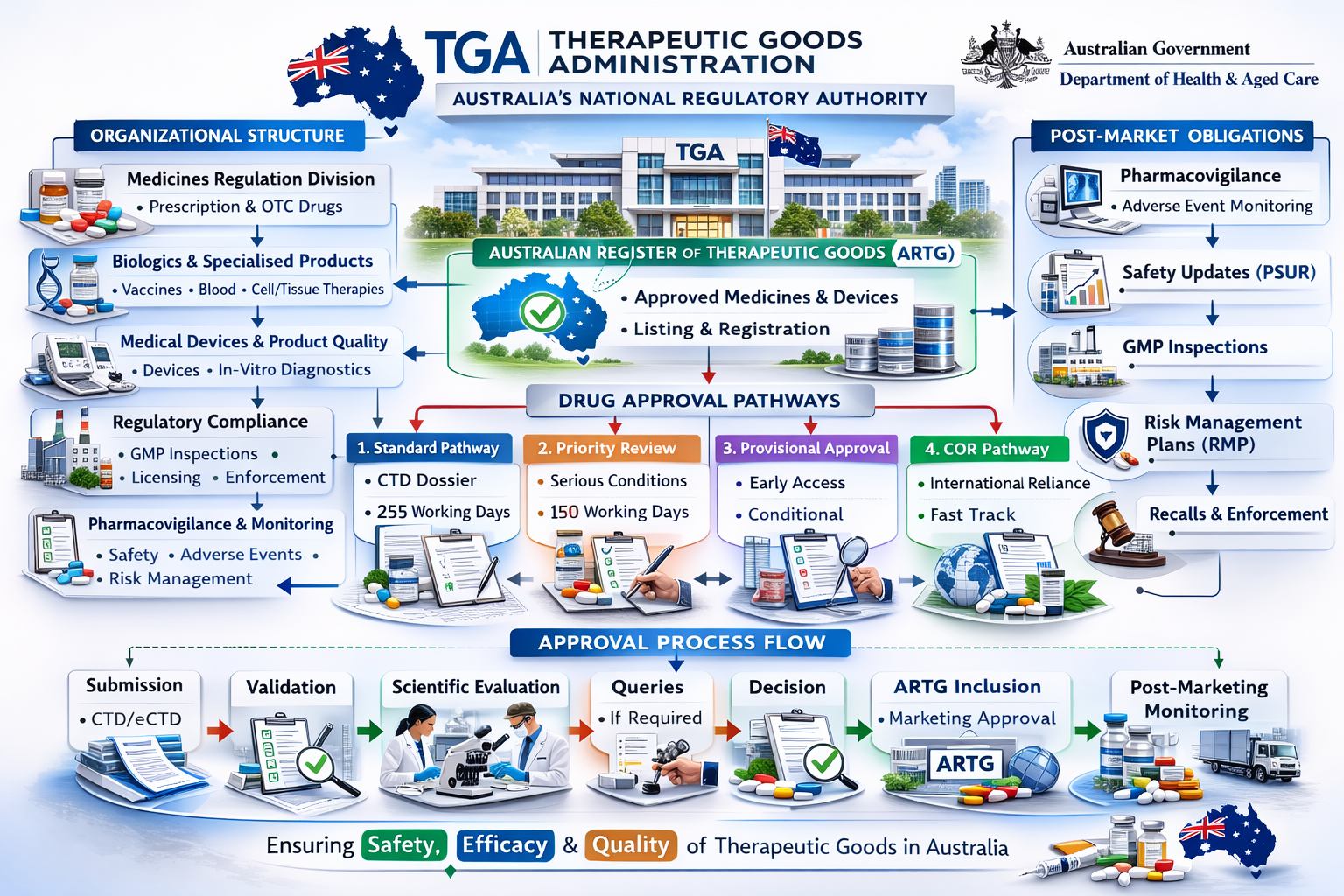 Organizational Structure
Organizational Structure
TGA functions through specialized divisions responsible for evaluation, compliance, and post-market monitoring.
Core Divisions
| Division | Key Responsibilities |
|---|---|
| Medicines Regulation Division | Evaluates prescription and over-the-counter medicines; reviews clinical, nonclinical, and quality data; grants marketing approvals |
| Biologics and Specialised Products | Regulates biological products including vaccines, blood products, and human tissue-based therapies; ensures manufacturing compliance |
| Medical Devices and Product Quality Division | Regulates medical devices and in-vitro diagnostics; conducts conformity assessments; oversees product quality standards |
| Regulatory Compliance Branch | Conducts GMP inspections; licenses manufacturers; investigates regulatory breaches; manages recalls and enforcement actions |
| Pharmacovigilance and Monitoring | Monitors adverse event reporting; evaluates post-approval benefit–risk profile; publishes safety alerts |
Australian Drug Approval System
In Australia, therapeutic goods must be included in the Australian Register of Therapeutic Goods (ARTG) before they can be legally supplied.
Australian Register of Therapeutic Goods (ARTG)
| Feature | Description |
|---|---|
| Central Database | Contains all approved therapeutic goods |
| Legal Requirement | Medicines and medical devices must be listed or registered before supply |
Pathways for Prescription Medicine Approval
| Pathway | Description | Timeline |
|---|---|---|
| Standard Pathway | Full evaluation of quality, safety, and efficacy; complete CTD submission; comprehensive scientific assessment | ~255 working days |
| Priority Review Pathway | For medicines addressing serious or life-threatening conditions | ~150 working days |
| Provisional Approval Pathway | Early approval based on preliminary clinical data; confirmatory data required later; time-limited registration | Conditional duration |
| Comparable Overseas Regulator (COR) Pathway | Relies on approval from trusted international regulators; abbreviated assessment | Shortened timeline |
Generic Drug Approval
| Requirement | Details |
|---|---|
| Bioequivalence Studies | Demonstration of equivalence with reference product |
| Quality & Stability Data | Full CMC data required |
| Regulatory Outcome | Inclusion in ARTG upon approval |
Post-Marketing Obligations
| Requirement | Description |
|---|---|
| Pharmacovigilance Reporting | Continuous adverse event monitoring |
| Periodic Safety Updates | Ongoing safety data submission |
| GMP Compliance | Routine inspections and regulatory oversight |
| Risk Management Plans (RMPs) | Risk mitigation and safety management strategies |
Process Flow Summary
Submission → Validation → Scientific Evaluation → Queries (if applicable) → Decision → ARTG Inclusion → Post-Marketing Monitoring
The TGA operates a structured, science-based regulatory framework ensuring that therapeutic goods available in Australia consistently meet rigorous standards of safety, quality, and effectiveness throughout their lifecycle.
