MHRA Approval System
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
Overview
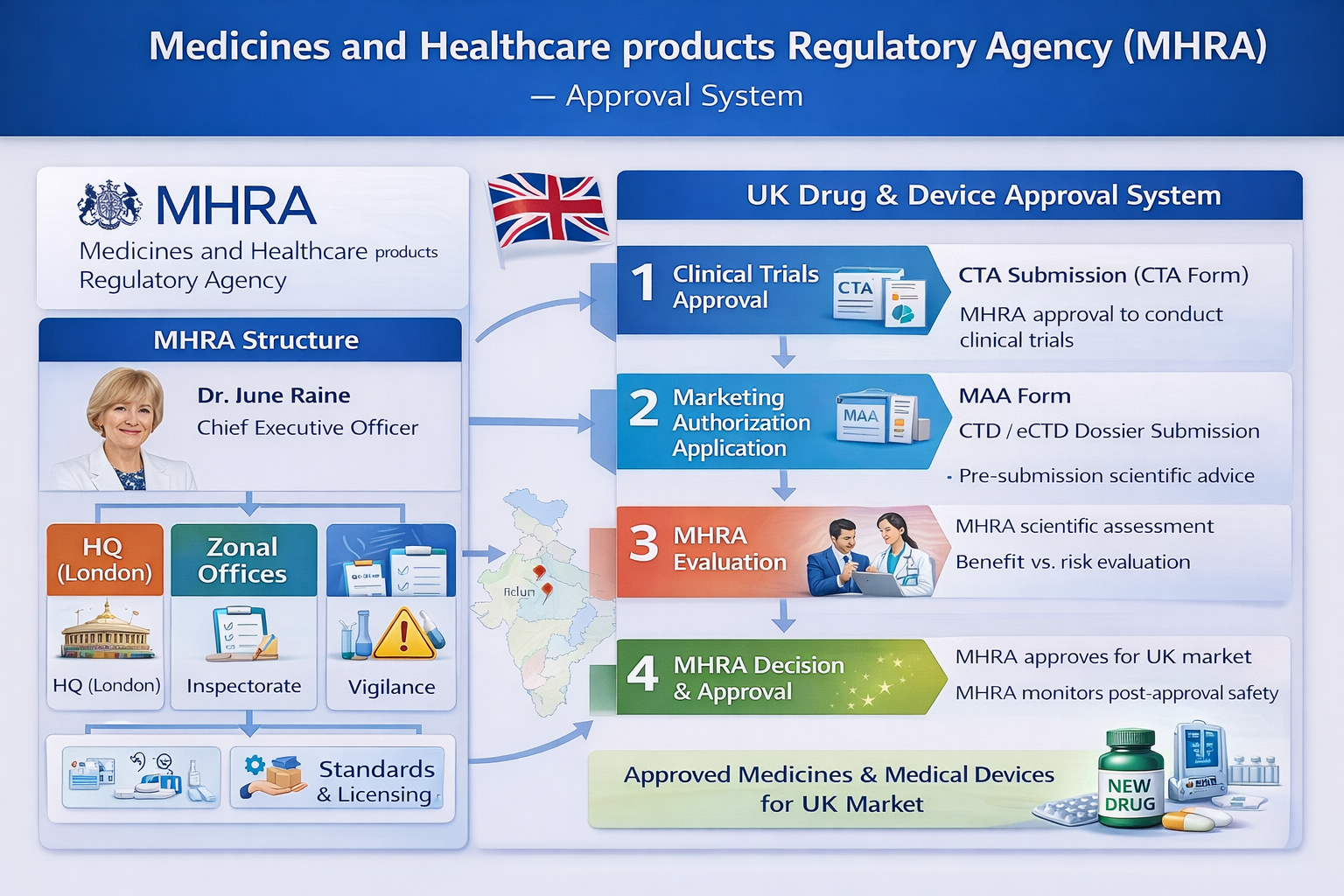
The Medicines and Healthcare products Regulatory Agency (MHRA) is the regulatory authority responsible for medicines and medical devices in the United Kingdom. Its primary role is to ensure that products meet the required standards of safety, quality, and efficacy before they are approved for use and while they remain on the market. The MHRA operates under the Department of Health and Social Care and functions independently from the European Medicines Agency following Brexit.
The agency uses a science-based regulatory framework that combines product evaluation, inspections, and post-marketing surveillance to protect public health across the United Kingdom.
MHRA Organizational Structure
The MHRA is led by the Chief Executive Officer, who is responsible for overall regulatory strategy and operational management. Supporting the CEO is the Executive Committee, which oversees policy implementation, scientific reviews, and enforcement activities. In addition, independent advisory committees provide external scientific expertise to support complex regulatory decisions and ensure objectivity.
The agency is organized into major divisions, each responsible for specific regulatory functions.
| Division | Core Function |
|---|---|
| Licensing Division | Reviews Marketing Authorization Applications, including clinical, nonclinical, and quality data |
| Vigilance and Risk Management Division | Monitors post-marketing safety and manages risk minimization activities |
| Inspection, Enforcement and Standards Division | Conducts GMP, GCP, and GDP inspections and ensures regulatory compliance |
| Devices Division | Regulates medical devices and in vitro diagnostic products |
MHRA Marketing Authorization Routes
The MHRA provides multiple pathways for obtaining marketing authorization, depending on the type of product and its regulatory history.
| Route | Description | Applicability |
|---|---|---|
| National Procedure | Direct submission to MHRA with full scientific assessment | Valid in Great Britain (England, Scotland, Wales) |
| International Recognition Procedure (IRP) | Abbreviated review for products approved by trusted regulators such as EU or US authorities | Accelerated approval pathway |
| Decentralized & Mutual Recognition (Legacy) | Based on former EU procedures; used in certain Northern Ireland arrangements | Limited post-Brexit applicability |
Step-by-Step MHRA Approval Process
| Step | Process Description |
|---|---|
| Step 1 – Submission | Marketing Authorization Application submitted via MHRA portal in CTD/eCTD format |
| Step 2 – Validation | Administrative check for completeness and confirmation of procedure |
| Step 3 – Scientific Assessment | Approximately 210-day review of quality, safety, and efficacy; queries may be issued with clock-stop periods |
| Step 4 – Benefit–Risk Decision | Positive or negative decision based on scientific evaluation |
| Step 5 – Post-Approval Obligations | Implementation of risk management plans, safety reporting, and inspections |
Accelerated Pathways
| Pathway | Purpose |
|---|---|
| Innovative Licensing and Access Pathway (ILAP) | Provides early regulatory support for innovative medicines |
| Conditional Marketing Authorization | Allows early approval based on limited data with defined obligations |
Process Flow Summary
| Sequence | Regulatory Stage |
|---|---|
| 1 | Submission |
| 2 | Validation |
| 3 | Scientific Review (approximately 210 days) |
| 4 | Queries and Company Response |
| 5 | Benefit–Risk Decision |
| 6 | Marketing Authorization |
The MHRA operates through a structured and science-driven regulatory system. It provides national approval pathways and international recognition mechanisms to ensure timely access to medicines while maintaining high standards of patient safety and product quality within the United Kingdom.