Annual Reports
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
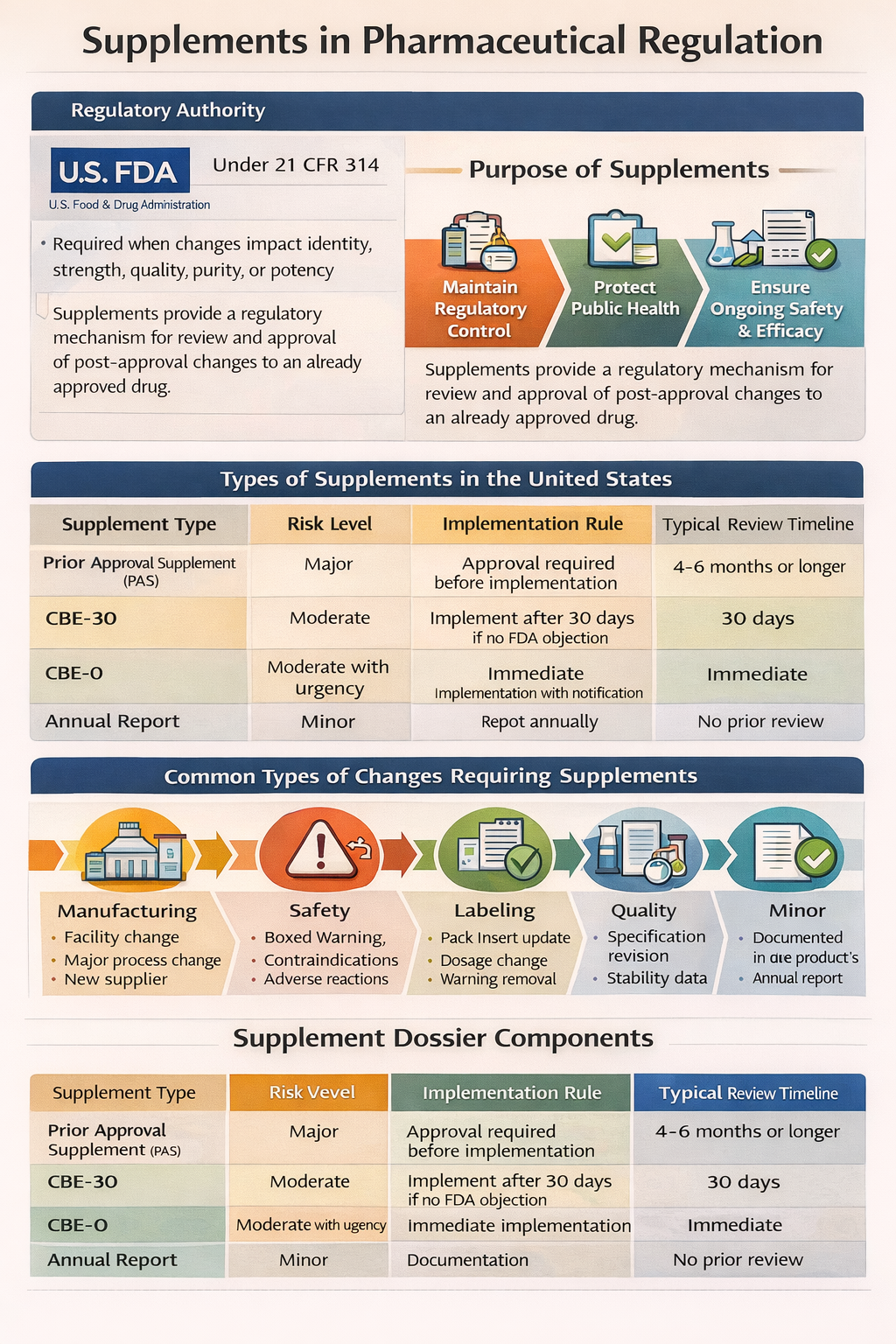
Annual Reports are mandatory post-approval regulatory submissions required for approved drug applications in the United States. They apply to New Drug Applications, Abbreviated New Drug Applications, and certain Biologics License Applications. The requirement is established under 21 CFR 314.81 and is enforced by the U.S. Food and Drug Administration.
An Annual Report must be submitted within 60 days of the anniversary date of the original approval of the application. The obligation continues as long as the product remains approved, even if it is not actively marketed.
Regulatory Purpose
The Annual Report serves as a structured compliance mechanism that allows the authority to monitor low-risk post-approval changes that were implemented without prior approval. It ensures transparency, regulatory oversight, and documentation of continued product quality and compliance.
Unlike supplements, Annual Reports are not submitted for review before implementation of changes. They are retrospective notifications covering changes made during the reporting period.
Scope and Applicability
Annual Reports are required for:
Approved NDA products
Approved ANDA products
Certain BLAs regulated under drug regulations
They do not apply to products that have been formally withdrawn or transferred.
Submission Timeline
| Requirement | Details |
|---|---|
| Reporting Period | 12 months from last anniversary date |
| Submission Deadline | Within 60 days after anniversary |
| Format | Electronic submission in eCTD format |
| Frequency | Every year for each approved application |
Content Structure of an Annual Report
An Annual Report must include specific regulatory elements. The submission generally contains the following structured components.
| Section | Description |
|---|---|
| Cover Letter | Identifies application number and reporting period |
| Summary of Changes | Overview of minor changes implemented |
| CMC Changes | Description of annual reportable quality changes |
| Stability Data | Summary of ongoing stability studies |
| Labeling Changes | Minor editorial updates if applicable |
| Post-Marketing Commitments | Status of required commitments |
| Distribution Data | Information on product distribution if required |
Types of Changes Reported
Only changes with minimal potential to adversely affect product quality, safety, or efficacy may be included in an Annual Report. Examples include:
Editorial labeling corrections
Minor equipment replacement of identical design
Tightening of specifications
Minor container closure adjustments
Non-critical manufacturing process optimizations
Changes that could significantly impact product quality or safety must be submitted separately as Prior Approval Supplements or CBE submissions and cannot be downgraded to annual reporting.
Regulatory Classification Comparison
| Change Category | Submission Type | Implementation Rule |
|---|---|---|
| Major Change | Prior Approval Supplement | Approval before implementation |
| Moderate Change | CBE-30 or CBE-0 | 30 days or immediate implementation |
| Minor Change | Annual Report | Implement first, report annually |
Stability Reporting Requirements
Ongoing stability data must be summarized in the Annual Report. This confirms that the product continues to meet approved specifications throughout its shelf life. Any out-of-specification results must be properly investigated and documented.
Labeling in Annual Reports
Minor editorial labeling corrections such as typographical fixes may be included. However, safety-related labeling changes cannot be deferred to Annual Reports and must follow appropriate supplement pathways.
Inspection and Audit Perspective
During regulatory inspections, inspectors verify:
Timely submission of Annual Reports
Correct classification of reported changes
Consistency between implemented changes and reported documentation
Adequacy of stability data
Accurate documentation of manufacturing updates
Failure to submit Annual Reports or misclassification of higher-risk changes as annual reportable can lead to compliance observations or warning letters.
Relationship with Lifecycle Management
Annual Reports form part of the post-approval lifecycle framework. They complement supplements and pharmacovigilance reporting by ensuring minor changes are documented systematically. Together, these mechanisms maintain continuous regulatory oversight without requiring a full re-approval process.
Comparison with European Union
In the European Union regulatory system coordinated by the European Medicines Agency, post-approval changes are handled through the variation system. There is no direct equivalent to the U.S. Annual Report system for minor changes. Instead, even small changes may require Type IA notification.
Compliance Risk Areas
Delayed submission beyond the 60-day window
Failure to report eligible minor changes
Improper downgrade of moderate or major changes
Incomplete stability summaries
Inadequate documentation of manufacturing updates
Strategic Importance
Annual Reports ensure ongoing transparency with the regulator while allowing operational flexibility for low-risk improvements. They represent a balance between regulatory oversight and efficient product lifecycle management.
In regulatory affairs practice, understanding the distinction between Annual Reportable changes and supplement-requiring changes is critical for compliance, inspection readiness, and proper change control classification.
