Introduction to Regulatory Affairs Function
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
Overview
Regulatory Affairs (RA) is a critical function within the pharmaceutical, biotechnology, and medical device industries. It ensures that products are developed, manufactured, tested, approved, and marketed in accordance with applicable laws, regulations, and quality standards. The main objective of Regulatory Affairs is to protect public health by ensuring that medicines and medical products are safe, effective, and of high quality before they reach patients.
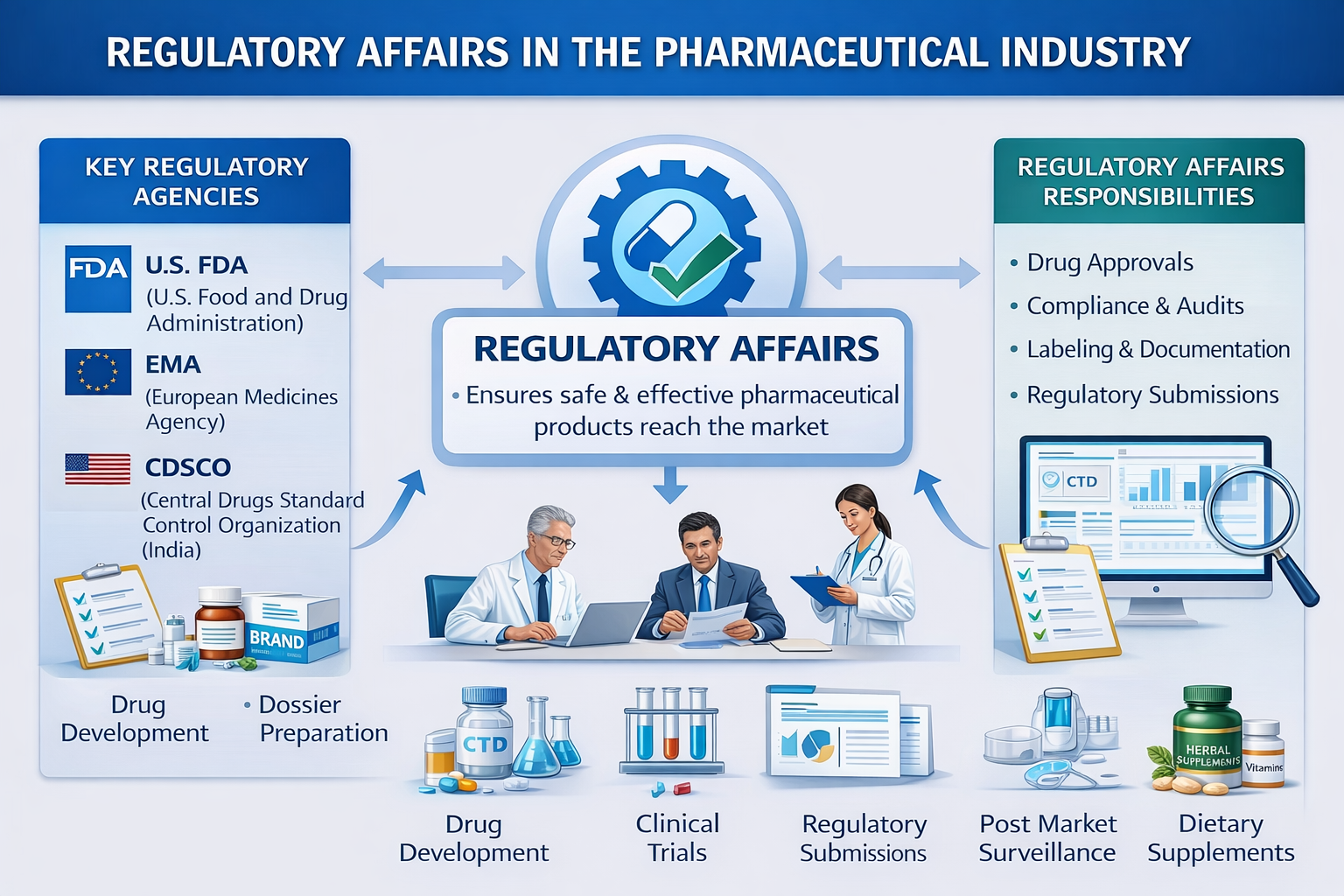
Regulatory professionals act as the bridge between companies and health authorities such as the U.S. Food and Drug Administration, the European Medicines Agency, and the Central Drugs Standard Control Organization. They interpret regulations, prepare submissions, and manage communication with regulatory agencies throughout the product lifecycle.
The Regulatory Affairs function is involved from the early research stage of a product and continues through clinical trials, product approval, and post-marketing activities. This lifecycle involvement ensures continuous compliance and long-term product success.
Core Objectives of Regulatory Affairs
| Objective | Strategic Purpose |
|---|---|
| Regulatory Compliance | Ensure adherence to national and international laws and guidelines |
| Timely Product Approval | Support efficient and successful market authorization |
| License Maintenance | Manage product registrations, renewals, and variations |
| Public Health Protection | Ensure safety, efficacy, and quality of products |
| Global Market Access | Enable entry into multiple regulatory regions |
| Risk Management | Identify and reduce regulatory and compliance risks |
These objectives help organizations maintain compliance while also supporting business continuity and market growth.
Role of Regulatory Affairs Across the Product Lifecycle
Regulatory Affairs plays a strategic and operational role at every stage of product development and commercialization.
Early Development Stage
| Activity | Function |
|---|---|
| Regulatory Strategy Planning | Define development and approval pathway |
| Regulatory Classification Guidance | Determine product category and requirements |
| Preclinical Advisory | Guide study design and compliance standards |
| IND Support | Assist in Investigational New Drug submission |
At this stage, Regulatory Affairs ensures that research and development activities align with regulatory expectations, reducing the risk of delays or rejections later.
Clinical Development Stage
| Activity | Function |
|---|---|
| Clinical Protocol Review | Ensure regulatory and ethical compliance |
| Clinical Trial Application Submission | Obtain authorization for trials |
| GCP Compliance Oversight | Maintain adherence to Good Clinical Practice |
| Agency Communication | Manage interactions with regulators |
During clinical trials, Regulatory Affairs ensures that studies follow ethical, scientific, and regulatory requirements.
Marketing Authorization Stage
| Activity | Function |
|---|---|
| NDA/MAA Preparation | Compile complete regulatory submission dossiers |
| Safety, Efficacy, and Quality Documentation | Consolidate scientific and technical data |
| Regulatory Query Management | Address authority questions and deficiencies |
| Inspection Coordination | Prepare for audits and facility reviews |
At this stage, Regulatory Affairs leads the preparation and submission of major applications required for product approval.
Post-Marketing Stage
| Activity | Function |
|---|---|
| Variation and Label Management | Update product information as required |
| Pharmacovigilance Compliance | Monitor adverse events and safety data |
| Periodic Safety Reporting | Submit mandatory safety updates |
| Lifecycle Management | Support renewals and product changes |
Post-approval responsibilities ensure ongoing compliance and continued patient safety.
Key Responsibilities of Regulatory Affairs Professionals
| Responsibility Area | Description |
|---|---|
| Dossier Preparation | Compile and submit regulatory applications |
| Regulatory Interpretation | Analyze evolving guidelines and policies |
| Authority Liaison | Act as primary contact with health agencies |
| GMP Compliance Oversight | Ensure manufacturing meets standards |
| Documentation Management | Maintain regulatory records and databases |
| Regulatory Intelligence | Monitor global regulatory changes |
| Audit and Inspection Support | Prepare for internal and external reviews |
Regulatory professionals combine scientific knowledge with legal, technical, and strategic expertise.
Regulatory Documentation and Submissions
| Submission Type | Purpose |
|---|---|
| Investigational New Drug (IND) | Authorization for human clinical trials |
| New Drug Application (NDA) | Approval to market new innovator drugs |
| Abbreviated New Drug Application (ANDA) | Approval pathway for generic drugs |
| Marketing Authorization Application (MAA) | European market approval |
| Drug Master File (DMF) | Confidential manufacturing and quality data |
| Common Technical Document (CTD/eCTD) | Standardized global submission format |
Standardized formats such as CTD and eCTD allow harmonized regulatory submissions across different countries.
Types of Regulatory Affairs
| Specialization | Focus Area |
|---|---|
| Pharmaceutical Regulatory Affairs | Small-molecule medicines |
| Biotechnology Regulatory Affairs | Biologics and advanced therapies |
| Medical Device Regulatory Affairs | Devices and diagnostics |
| Generic Drug Regulatory Affairs | Abbreviated approval pathways |
| International Regulatory Affairs | Multi-country registration strategies |
Each specialization requires knowledge of specific regulations, guidelines, and approval pathways.
Skills Required in Regulatory Affairs
| Skill Category | Competency |
|---|---|
| Regulatory Knowledge | Understanding of global regulations and guidelines |
| Clinical and Scientific Insight | Knowledge of drug development processes |
| Technical Writing | Preparation of regulatory documents |
| Analytical Thinking | Risk assessment and problem solving |
| Communication Skills | Interaction with regulators and teams |
| Legal Interpretation | Understanding regulatory laws and frameworks |
Regulatory professionals must integrate science, quality systems, legal knowledge, and strategic planning.
Importance of Regulatory Affairs
| Impact Area | Benefit |
|---|---|
| Patient Safety | Ensures safe and effective medicines |
| Risk Mitigation | Prevents product rejection or recall |
| Market Expansion | Supports global product launches |
| Compliance Assurance | Maintains ethical and legal standards |
| Corporate Reputation | Builds regulatory credibility |
Regulatory Affairs plays a vital role in ensuring that healthcare products meet strict regulatory standards while supporting innovation, market access, and patient safety.
