Submission Readiness
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
Overview
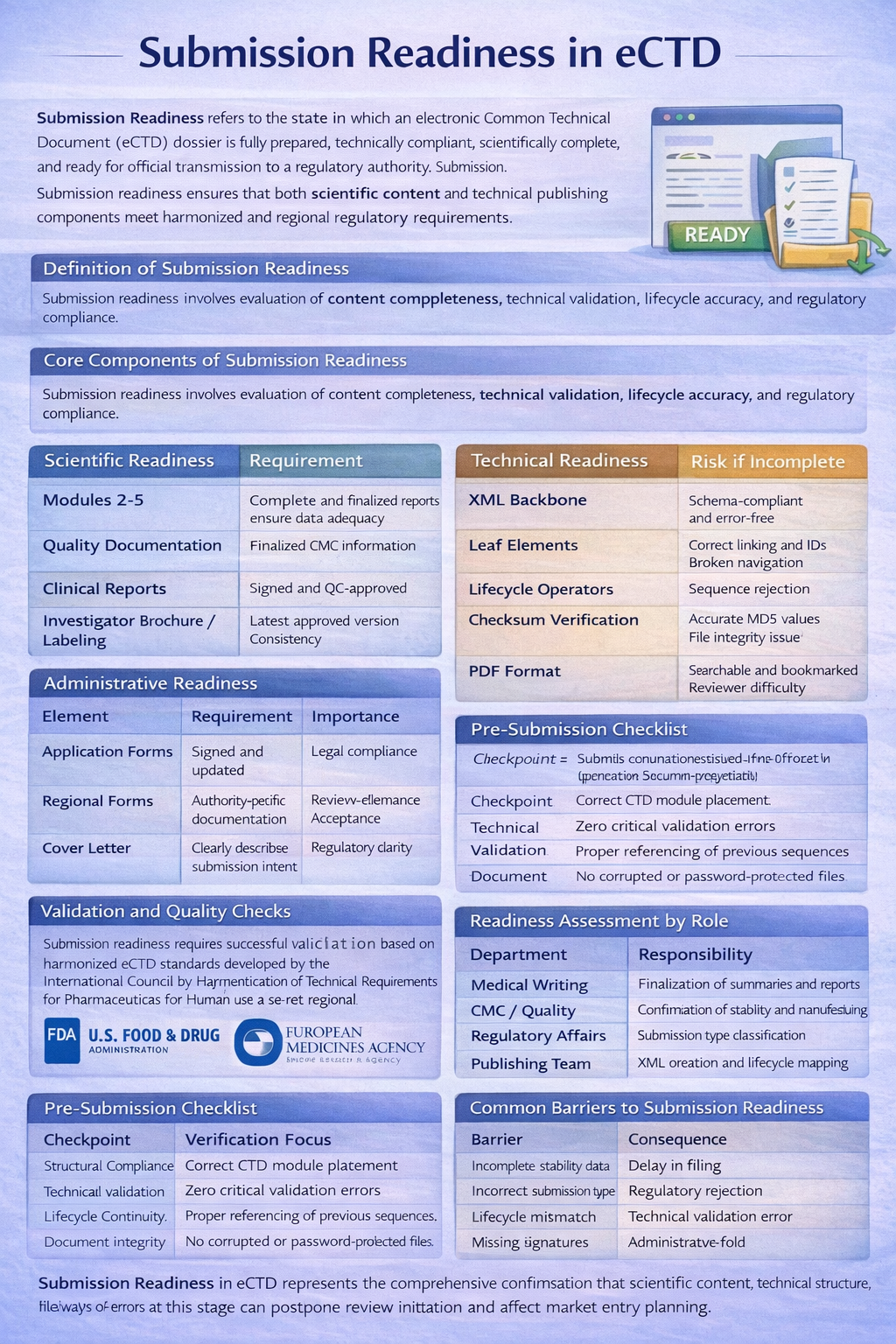
Submission Readiness refers to the state in which an eCTD dossier is fully prepared, technically compliant, scientifically complete, and ready for official transmission to a regulatory authority. It represents the final checkpoint before submission via the agency’s electronic gateway.
Submission readiness ensures that both scientific content and technical publishing components meet harmonized and regional regulatory requirements.
Definition of Submission Readiness
| Parameter | Description |
|---|---|
| Scope | Covers scientific, technical, and administrative completeness |
| Stage | Final phase before submission |
| Responsibility | Regulatory Affairs, Medical Writing, Publishing, QA |
| Outcome | Dossier ready for gateway transmission |
Core Components of Submission Readiness
Submission readiness involves evaluation of content completeness, technical validation, lifecycle accuracy, and regulatory compliance.
Scientific Readiness
| Element | Requirement | Purpose |
|---|---|---|
| Modules 2–5 | Complete and finalized reports | Ensure data adequacy |
| Quality Documentation | Finalized CMC information | Compliance with specifications |
| Clinical Reports | Signed and QC-approved | Accuracy and reliability |
| Investigator Brochure / Labeling | Latest approved version | Consistency |
Technical Readiness
| Element | Requirement | Risk if Incomplete |
|---|---|---|
| XML Backbone | Schema-compliant and error-free | Validation failure |
| Leaf Elements | Correct linking and IDs | Broken navigation |
| Lifecycle Operators | Properly mapped | Sequence rejection |
| Checksum Verification | Accurate MD5 values | File integrity issue |
| PDF Format | Searchable and bookmarked | Reviewer difficulty |
Administrative Readiness
| Element | Requirement | Importance |
|---|---|---|
| Application Forms | Signed and updated | Legal compliance |
| Regional Forms | Authority-specific documentation | Acceptance criteria |
| Cover Letter | Clearly describing submission intent | Regulatory clarity |
| Fee Payment Confirmation | Proof of payment | Administrative approval |
Validation and Quality Checks
Submission readiness requires successful validation based on harmonized eCTD standards developed by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use and regional technical specifications.
Regulatory authorities such as the U.S. Food and Drug Administration and the European Medicines Agency perform automated validation before initiating scientific review.
Pre-Submission Checklist
| Checkpoint | Verification Focus |
|---|---|
| Structural Compliance | Correct CTD module placement |
| Technical Validation | Zero critical validation errors |
| Lifecycle Continuity | Proper referencing of previous sequences |
| Document Integrity | No corrupted or password-protected files |
| Completeness | All mandatory sections included |
Readiness Assessment by Role
| Department | Responsibility |
|---|---|
| Medical Writing | Finalization of summaries and reports |
| CMC / Quality | Confirmation of stability and manufacturing data |
| Regulatory Affairs | Submission type classification |
| Publishing Team | XML creation and lifecycle mapping |
| Quality Assurance | Independent compliance verification |
Common Barriers to Submission Readiness
| Barrier | Consequence |
|---|---|
| Incomplete stability data | Delay in filing |
| Incorrect submission type | Regulatory rejection |
| Lifecycle mismatch | Technical validation error |
| Missing signatures | Administrative hold |
Strategic Importance
Submission readiness directly impacts regulatory timelines, review clock initiation, and product approval pathways. Delays or errors at this stage can postpone review initiation and affect market entry planning.
Conclusion
Submission Readiness in eCTD represents the comprehensive confirmation that scientific content, technical structure, XML backbone, lifecycle mapping, and administrative documentation are fully compliant and validated. It is the final quality gate before regulatory transmission and plays a critical role in ensuring successful acceptance and timely review of pharmaceutical applications.
