Overview of Regulatory Submissions
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
What are Regulatory Submissions?
Introduction
Regulatory submissions are formal applications and structured data packages that pharmaceutical companies submit to regulatory authorities to obtain approval for clinical trials, marketing authorization, or post-approval changes. These submissions contain documented scientific evidence demonstrating that a drug is safe, effective, and manufactured according to established quality standards.
They are reviewed by regulatory agencies such as the U.S. Food and Drug Administration, the European Medicines Agency, and the Central Drugs Standard Control Organization. Regulatory submissions form the foundation of regulatory decision-making and are essential for gaining and maintaining market authorization.
Purpose of Regulatory Submissions
| Purpose | Regulatory Objective |
|---|---|
| Clinical Trial Authorization | Obtain approval to conduct human studies |
| Marketing Approval | Secure authorization for product commercialization |
| Generic Drug Approval | Demonstrate equivalence to a reference product |
| Post-Approval Changes | Notify and obtain approval for product modifications |
| Lifecycle Maintenance | Maintain registration and regulatory compliance |
| Patient Safety Assurance | Ensure quality, safety, and efficacy standards |
Regulatory submissions promote transparency, accountability, and scientific validation throughout product development and commercialization.
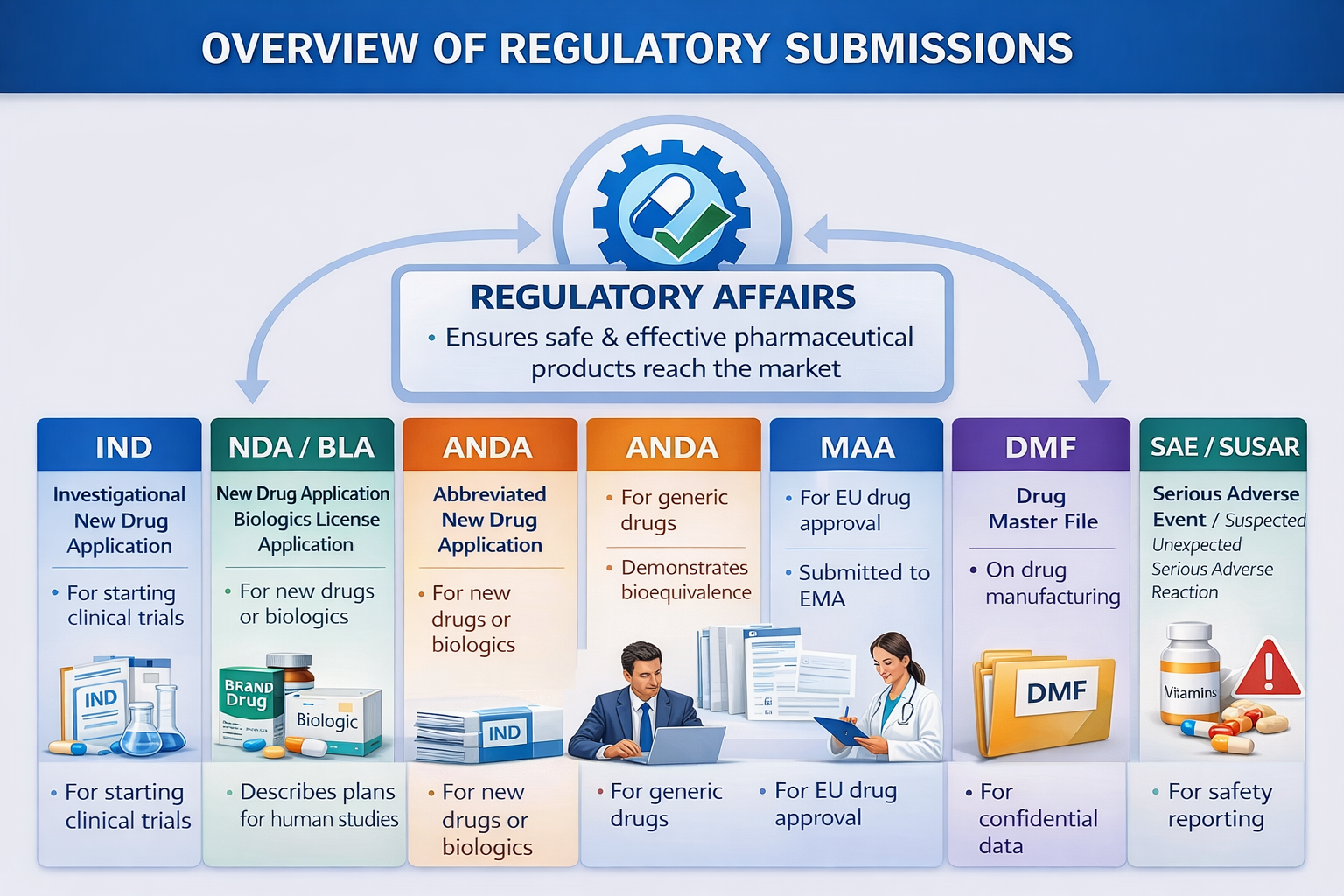
Major Types of Regulatory Submissions
| Submission Type | Purpose | Key Components |
|---|---|---|
| Investigational New Drug (IND) Application | Permission to begin human clinical trials | Preclinical data, CMC information, clinical protocols |
| New Drug Application (NDA) | Marketing approval for innovator drugs | Full nonclinical, clinical, and manufacturing data with labeling |
| Abbreviated New Drug Application (ANDA) | Approval for generic drugs | Bioequivalence data and quality documentation |
| Marketing Authorization Application (MAA) | European Union marketing approval | Complete quality, safety, and efficacy data |
| Drug Master File (DMF) | Confidential manufacturing information | Drug substance and facility details |
| Supplemental/Variation Submission | Post-approval modifications | Manufacturing, labeling, safety, or indication updates |
Each submission type serves a specific regulatory purpose and must meet defined scientific and legal requirements.
Structure of Regulatory Submissions (CTD Format)
Most global regulatory authorities follow the Common Technical Document (CTD) structure, which standardizes submission formats and simplifies international approvals.
| CTD Module | Content |
|---|---|
| Module 1 – Administrative Information | Regional administrative forms and labeling |
| Module 2 – Summaries and Overviews | Quality, nonclinical, and clinical summaries |
| Module 3 – Quality (CMC) | Drug substance, formulation, manufacturing, and stability data |
| Module 4 – Nonclinical Reports | Pharmacology and toxicology study reports |
| Module 5 – Clinical Reports | Phase I–III clinical study data and statistical analyses |
The CTD format promotes consistency, efficiency, and streamlined review across different regulatory regions.
Electronic Submissions
| Digital Component | Function |
|---|---|
| Electronic Common Technical Document (eCTD) | Standardized electronic submission format |
| Online Submission Portals | Secure digital filing with regulatory authorities |
| Document Management Systems | Centralized tracking and storage of regulatory files |
| Version Control Systems | Lifecycle tracking of submission updates |
Electronic submissions improve traceability, transparency, and overall review efficiency.
Regulatory Review Process
| Review Stage | Activity |
|---|---|
| Administrative Review | Check submission completeness |
| Scientific Evaluation | Assess quality, safety, and efficacy data |
| Facility Inspection | Verify GMP compliance at manufacturing sites |
| Query Management | Address deficiency letters and authority questions |
| Final Decision | Approval, rejection, or request for additional data |
This structured review process ensures a comprehensive scientific and regulatory assessment before market authorization.
Importance of Regulatory Submissions
| Impact Area | Benefit |
|---|---|
| Scientific Validation | Supports evidence-based regulatory decisions |
| Patient Protection | Ensures drug safety and effectiveness |
| Market Access | Enables product commercialization |
| Compliance Assurance | Meets evolving regulatory standards |
| Global Registration | Facilitates international approvals |
Regulatory submissions represent comprehensive scientific documentation that demonstrates a product’s safety, efficacy, and quality. Proper planning, accurate preparation, and strong regulatory strategy are essential for achieving timely approval and sustainable market access.
