DMF Types
Join our community on Telegram!
Join the biggest community of Pharma students and professionals.
DMF & Its Type?
A Drug Master File (DMF) is a confidential submission made to the U.S. Food and Drug Administration that provides detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storage of human drug products. A DMF allows a manufacturer to protect proprietary information while authorizing its reference in regulatory submissions such as INDs, NDAs, or ANDAs. DMFs are governed under 21 CFR 314.420 and serve as a key regulatory mechanism in supply chain management and contract manufacturing environments.
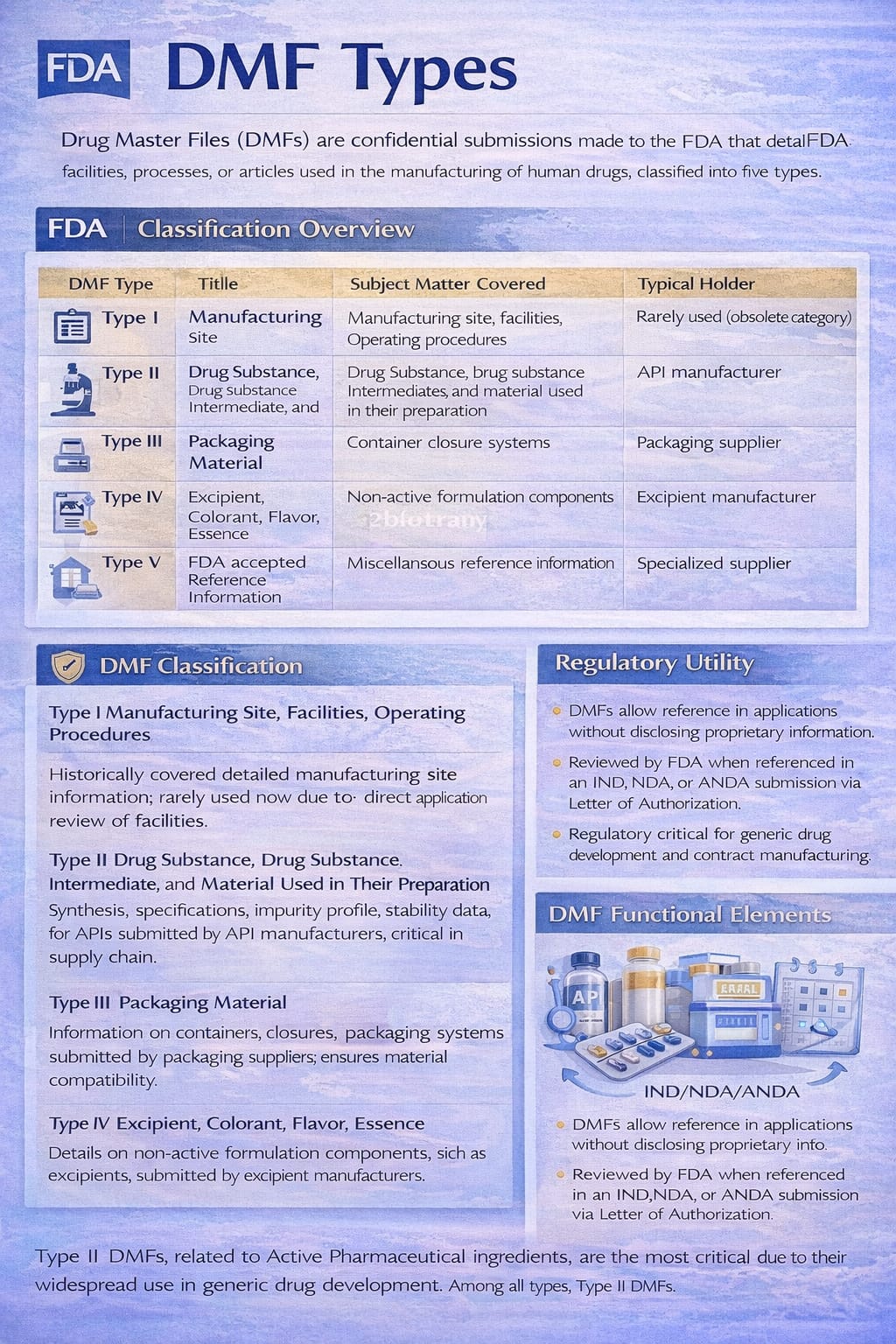
The FDA classifies DMFs into five types based on the nature of the information submitted. Each type serves a specific regulatory purpose.
DMF Classification Overview
| DMF Type | Title | Subject Matter Covered | Typical Holder |
|---|---|---|---|
| Type I | Manufacturing Site, Facilities, Operating Procedures | Manufacturing site details | Rarely used (obsolete category) |
| Type II | Drug Substance, Drug Substance Intermediate, and Material Used in Their Preparation | Active Pharmaceutical Ingredient (API) | API manufacturer |
| Type III | Packaging Material | Container closure systems | Packaging supplier |
| Type IV | Excipient, Colorant, Flavor, Essence | Non-active formulation components | Excipient manufacturer |
| Type V | FDA Accepted Reference Information | Miscellaneous reference information | Specialized supplier |
Type I DMF
Type I DMFs historically covered manufacturing site, facilities, and operating procedures. However, the FDA no longer accepts Type I DMFs. Facility information is now typically reviewed directly within application submissions or through inspection processes. Therefore, Type I DMFs are considered obsolete for new submissions.
Type II DMF
Type II DMFs are the most common and commercially significant. They contain detailed information about drug substances, drug substance intermediates, and materials used in their preparation. This includes synthesis pathway, impurity profile, specifications, analytical methods, stability data, and manufacturing controls. Type II DMFs are typically submitted by Active Pharmaceutical Ingredient manufacturers who supply multiple finished product companies. The applicant referencing the DMF must obtain a Letter of Authorization (LOA) from the DMF holder.
Type III DMF
Type III DMFs contain information on packaging materials such as bottles, caps, blister packs, vial systems, and other container closure components. These submissions ensure that packaging materials meet regulatory standards for compatibility, stability, and product protection. Type III DMFs are typically submitted by packaging component manufacturers.
Type IV DMF
Type IV DMFs cover excipients, colorants, flavors, and other non-active components used in drug formulations. These DMFs provide manufacturing details, specifications, impurity information, and safety data for excipients. They are particularly useful when proprietary manufacturing processes are involved.
Type V DMF
Type V DMFs are used for FDA-accepted reference information that does not fall under Types II, III, or IV. These are less common and are used for unique materials or processes that require confidential submission.
Regulatory Utility of DMFs
DMFs are not approved or disapproved by the FDA. Instead, they are reviewed when referenced in an application such as an IND, NDA, or ANDA. The relationship between the DMF holder and the applicant is governed through a Letter of Authorization.
DMF Functional Elements
| Component | Purpose |
|---|---|
| Administrative Information | Identifies DMF holder and contact details |
| Quality Information | Detailed technical and manufacturing data |
| Letter of Authorization | Permits FDA to reference DMF in application |
| Annual Update | Maintains DMF active status |
Global Perspective
While DMFs are specific to the United States regulatory framework, similar confidential technical file systems exist globally. In Europe, Active Substance Master Files are used under the coordination of the European Medicines Agency, and in other jurisdictions similar mechanisms are implemented for confidential supplier documentation.
Career Perspective
For a Regulatory Affairs professional, especially in generic and API-focused companies, understanding Type II DMFs is essential. Key competencies include drafting Letters of Authorization, ensuring alignment between DMF data and application content, tracking DMF annual updates, responding to deficiency letters, and coordinating with API suppliers. In multinational pharmaceutical and CRO environments such as Pfizer, Novartis, Roche, and IQVIA, DMF management is an integral part of regulatory strategy and supply chain governance.
Conclusion
Drug Master Files are strategic regulatory tools that protect proprietary manufacturing information while enabling regulatory compliance across multiple applications. Among all types, Type II DMFs for active pharmaceutical ingredients are the most critical in generic drug development. Mastery of DMF classification, referencing mechanisms, and lifecycle management is essential for professionals working in global drug regulatory affairs.