How Long Does It Take to Get a Pharmacovigilance Job?
If you are planning to build a career in pharmacovigilance, one practical question always comes up:
How long does it actually take to get a pharmacovigilance job?
Is it possible in one month? Does it take six months? Or does it stretch to a year?
Let’s answer this clearly, realistically, and strategically — based on current hiring trends in India, global CRO expansion, and what truly determines your job conversion speed after pharmacovigilance training.
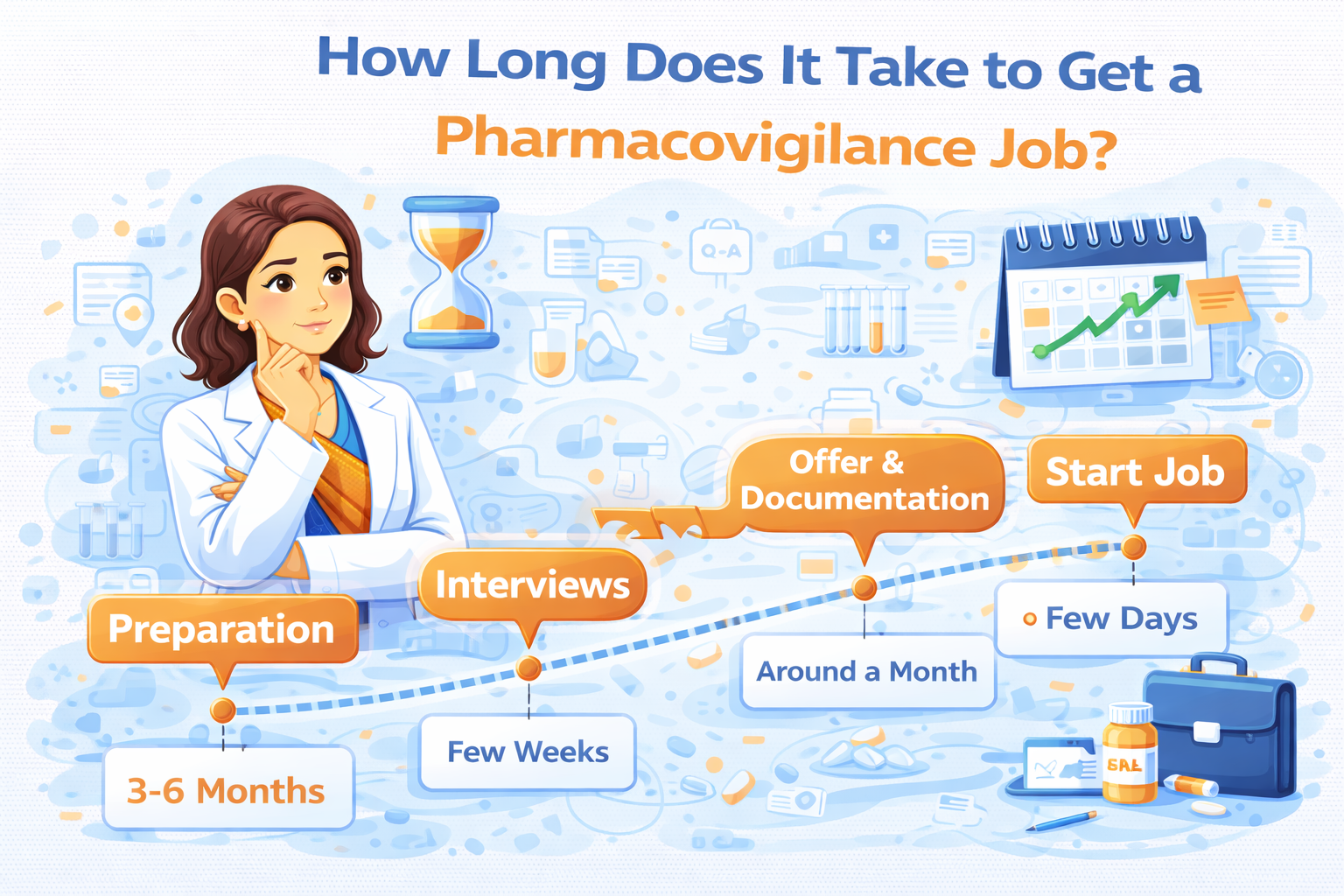
The Short Answer: 3–6 Months (For Most Serious Candidates)
For candidates who complete structured, job-oriented pharmacovigilance training and actively apply with the right strategy, the average timeline to secure a pharmacovigilance job is:
3–6 months
This includes:
-
Skill development
-
Resume preparation
-
Mock interview practice
-
Active applications
-
Interview rounds
-
Offer processing time
However, this timeline depends heavily on how you prepare.
Understanding the Hiring Landscape in Pharmacovigilance
Pharmacovigilance (PV) is the science of monitoring drug safety and reporting adverse events. With increasing regulatory requirements from agencies like USFDA and EMA, and growing outsourcing to India, the demand for trained drug safety professionals continues to rise.
Major organizations hiring for pharmacovigilance roles include:
-
IQVIA
-
Tata Consultancy Services
-
Accenture
-
Cognizant
These companies regularly recruit freshers and experienced candidates for roles like Drug Safety Associate, PV Associate, Safety Data Reviewer, and Case Processor.
But hiring is competitive. Companies prefer candidates who are immediately deployable — meaning they understand ICSR processing, regulatory timelines, and safety databases.
That is where structured pharmacovigilance training becomes critical.
Phase-Wise Breakdown of the Pharmacovigilance Job Timeline
Phase 1: Skill Development (1–2 Months)
The first step is completing structured pharmacovigilance training.
A proper training program must include:
-
ICSR case processing
-
Validity assessment
-
Seriousness criteria
-
Expectedness assessment
-
Causality evaluation
-
MedDRA coding basics
-
Narrative writing practice
-
Day 0 concept and reporting timelines
-
Introduction to Argus or ARIS workflows
Candidates who rely only on random YouTube videos or scattered materials often extend this phase to 6–12 months.
If you are serious about reducing your job timeline, structured pharmacovigilance training is non-negotiable.
You can explore a comprehensive, job-oriented pharmacovigilance training curriculum here:
https://thepharmadaily.com/pharmacovigilance-training
Phase 2: Resume & Interview Preparation (2–4 Weeks)
This is where many candidates underestimate the process.
After completing pharmacovigilance training, you must:
-
Create an ATS-friendly resume
-
Align resume with CRO job descriptions
-
Prepare technical answers for PV interviews
-
Practice case-based questions
-
Build confidence explaining ICSR workflows
Technical knowledge alone is not enough. You must communicate your understanding clearly and confidently.
Structured pharmacovigilance training that includes mock interviews significantly improves selection chances.
Phase 3: Application & Interview Cycle (1–3 Months)
Once you begin active job applications:
-
Resume shortlisting: 2–4 weeks
-
HR screening round
-
Technical round
-
Managerial round
-
Offer processing: 1–3 weeks
Sometimes you may attend multiple interviews before receiving your first offer. That is normal.
Consistency matters more than speed.
Candidates who apply strategically after completing structured pharmacovigilance training typically convert interviews within 2–3 months.
What Determines Whether It Takes 3 Months or 12 Months?
1. Quality of Pharmacovigilance Training
Not all pharmacovigilance training programs are designed for job readiness.
A strong training program must include:
-
Real case scenarios
-
Long narrative writing exercises
-
Regulatory reporting examples
-
Live case discussions
-
Interview question banks
-
Resume guidance
If your pharmacovigilance training is only theoretical, your interview confidence will suffer — and your timeline will increase.
Explore job-focused pharmacovigilance training here:
https://thepharmadaily.com/pharmacovigilance-training
2. Educational Background
Candidates from:
-
B.Pharm
-
M.Pharm
-
PharmD
-
BDS
-
BAMS
-
Life Sciences
can enter pharmacovigilance.
However, your degree alone does not guarantee speed. Your understanding of PV workflows does.
Strong pharmacovigilance training bridges that gap effectively.
3. Location Flexibility
Major hiring hubs in India include:
-
Pune
-
Bangalore
-
Hyderabad
-
Mumbai
Candidates open to relocation often get hired faster compared to those restricting themselves to one city.
4. Communication & Confidence
Many candidates know the concepts but struggle to explain:
-
What makes a case valid?
-
What is Day 0?
-
What qualifies as serious?
-
How do you draft a narrative?
-
What is the difference between listed and unlisted events?
This is why structured mock interviews inside pharmacovigilance training are essential.
Confidence shortens timelines.
Fast-Track Strategy: How to Get a Pharmacovigilance Job in 90 Days
If your goal is aggressive placement within 3 months, follow this roadmap:
Month 1:
Complete structured pharmacovigilance training
Practice 20–30 ICSR case scenarios
Master seriousness and validity criteria
Month 2:
Prepare resume
Attend mock interviews
Start applying daily to PV openings
Month 3:
Attend interviews
Improve based on feedback
Refine answers
Follow up strategically
Students who follow a disciplined, structured pharmacovigilance training pathway often secure roles within 3–6 months.
Why Some Candidates Take 1 Year
Common mistakes include:
-
Learning randomly without structured pharmacovigilance training
-
Avoiding mock interviews
-
Weak resume formatting
-
Fear of technical questions
-
Inconsistent applications
-
Lack of clarity about regulatory concepts
The delay is rarely due to lack of jobs. It is usually due to lack of structured preparation.
Is Pharmacovigilance Still a Good Career Choice in 2026?
Yes — and increasingly so.
Pharmacovigilance offers:
-
Regulatory-driven stability
-
Global career mobility
-
Structured career growth
-
Opportunities in aggregate reporting, signal detection, and risk management
-
Potential transition into regulatory affairs or medical writing
With increasing drug approvals and safety monitoring requirements worldwide, demand for trained professionals remains strong.
But the key is proper pharmacovigilance training.
If you are serious about entering this field with clarity and confidence, explore a complete job-oriented pharmacovigilance training program here:
https://thepharmadaily.com/pharmacovigilance-training
Final Verdict: How Long Does It Really Take?
Average timeline: 3–6 months
With structured pharmacovigilance training + resume optimization + interview preparation.
Can it happen faster? Yes — with focused effort and daily practice.
Can it take longer? Yes — without direction and structured learning.
Your timeline depends less on luck and more on preparation strategy.
If your goal is to secure a pharmacovigilance job this year, commit to structured pharmacovigilance training, build strong fundamentals, and prepare like a professional — not like a passive learner.
In competitive industries, clarity beats confusion.
And preparation beats uncertainty.



